There is a growing buzz around spectral process analytical technology (PAT) for the life sciences industry. Spectral PAT sounds really fancy. What is it, and how can it affect my company’s mission to produce treatments and cures that improve the quality of life for our patients?
Let’s start with the basics. The ultimate value proposition of every life sciences manufacturer is to produce goods that dramatically improve health and the corresponding quality of life. To fulfill this value proposition, a product must move through the value stream at a pace consistent with the demand for that given product. Before the product can be distributed to customers, it must be proven to be of sufficient quality as defined by that product’s regulatory filings.
Traditionally, product quality has been enforced using offline measurements and analysis. In-line measurement has been deployed where possible to enforce quality requirements for physical attributes like mass, temperature, pH, and conductivity, leaving more complex measurements like purity and potency for offline analysis. Therein lies the opportunity.
In-line spectroscopy has the potential to unlock product quality measurements that were previously limited to quality control labs. The benefits of such in-line analysis include enabling real-time product release, reducing labor requirements for production and release, and allowing for real-time process control in response to material input variability. This past year, BioPhorum published several articles on the opportunities to pursue in-line monitoring to enable real-time release.
- How to maximize business value from process analytical technologies
- Operational vision: Adoption of in-line monitoring and real-time release
- How fully implemented ILM/RTR will be a quantum leap for industry
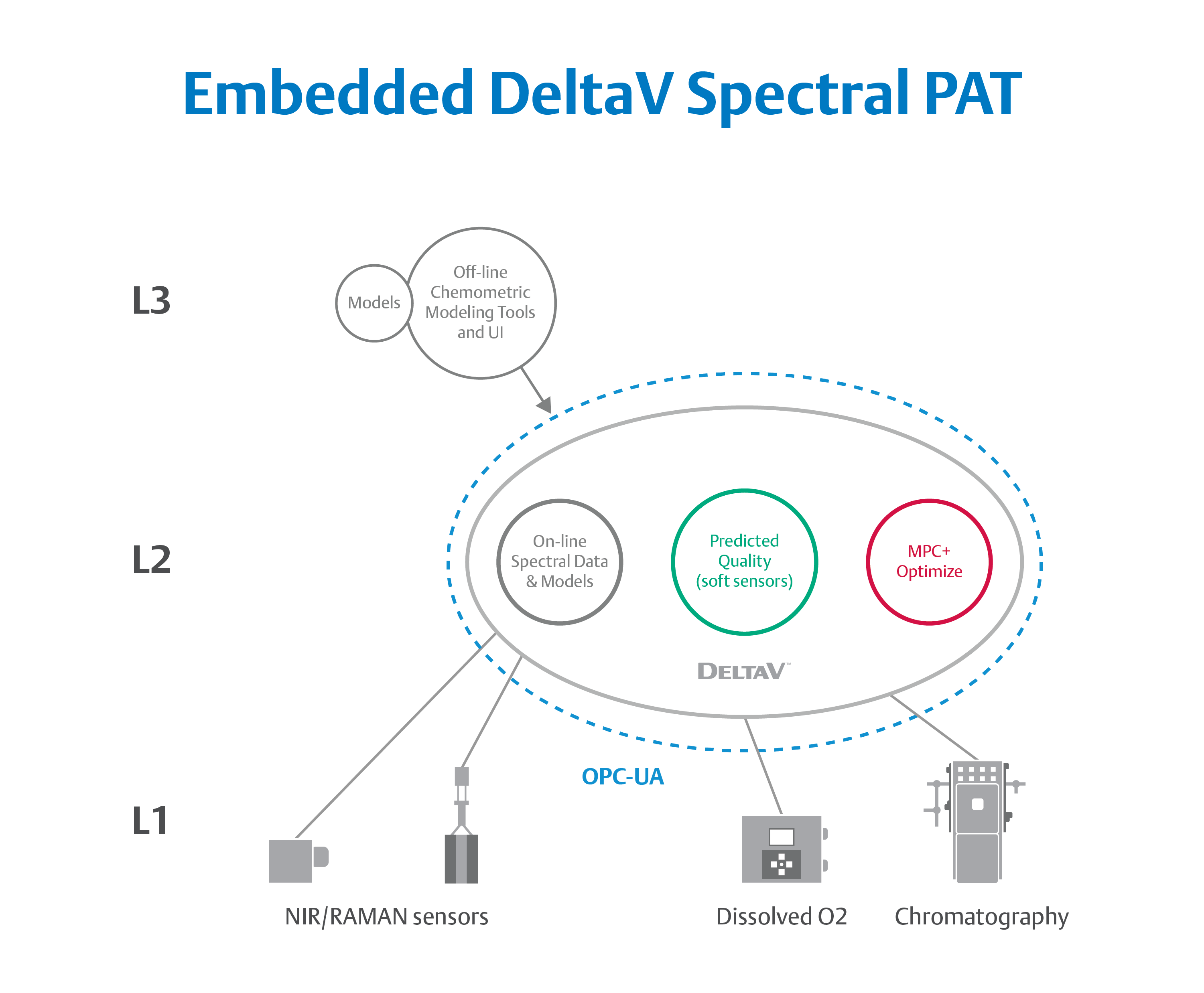
ILM components such as Emerson’s DeltaV Spectral PAT bring real-time quality testing into closed-loop control.
Because spectroscopic data contains a continuum of values across a range of wavelengths, frequencies, or frequency shifts, an advanced analytical system is required to process the values to determine conformance to desired quality attributes. To address this challenge, Emerson offers an integrated Spectral PAT solution for DeltaV, allowing manufacturers to pursue closed-loop control (and real-time quality assessments) using in-line spectroscopic instruments.
Last February, Emerson Automation Experts Todd Walden, Bob Lenich, and Bruce Greenwald published a blog post, Improve Speed to Market with Closed-Loop Process Control using Spectral PAT, highlighting several benefits of DeltaV Spectral PAT.
Spectral PAT offers a new approach to assessing quality attributes associated with purity and potency. To realize the full benefits of this technology, the industry will now be challenged to prove to regulatory agencies that process control and quality control rooted in real-time spectral analysis are as good or better than traditional lab-based analytical methods.
Now is an ideal time to re-think how critical quality attributes will be measured and enforced for products and processes currently under development. Companies that leverage in-line technologies like spectral PAT will have significant advantages regarding product lead times and costs.
Visit the DeltaV site to learn more about how Emerson can help you reduce lead time and improve quality using spectral PAT.