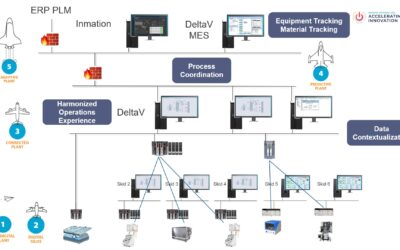
Emerson’s Bruce Greenwald, Christian Berg, and David Gray collaborated to present “Solutions for Advanced Therapeutics” at the Emerson Exchange 2025 Conference.
Christian Berg
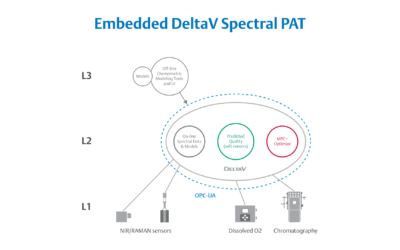
Real-World Implications of Spectral PAT for Life Sciences
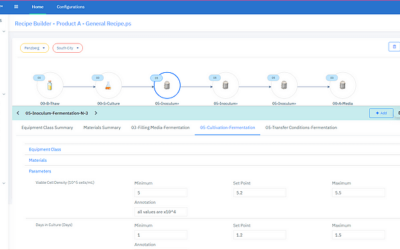
Emerson offers an integrated Spectral PAT solution for DeltaV, allowing manufacturers to pursue closed-loop control (and real-time quality assessments) using in-line spectroscopic instruments.
Manufacturing Solutions in Personalized Healthcare
Christian Berg describes some solutions and technologies which facilitate the pursuit of the adaptive plant for manufacturers in the Life Sciences industry, as defined by the BioPhorum Digital Plant Maturity Model.
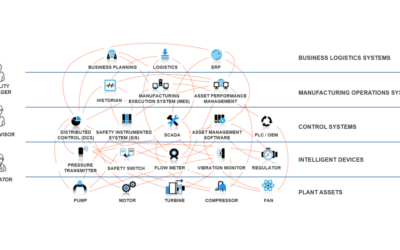
Adaptable Landscape Components Facilitate Digital Transformation
The BioPhorum Operations Group (BPOG) established a digital plant maturity model (DPMM) for biopharmaceutical manufacturing to define stages of factory evolution “from simple paper-based plants through to the fully automated and integrated ‘adaptive plant’ of the future.” The model gives organizations a common tool for industry comparison and measurement of progress. It also provides a platform to facilitate collaboration within the life sciences industry and its technology providers.
Overcoming Pharmaceutical and Biotech Manufacturing Challenges
Emerson’s Christian Berg presented Are We There Yet? Tackling Manufacturing Challenges with the Digital Plant at the Pharma Manufacturing World Summit in Boston.
Holistic Production Readiness Strategy is Key to Operational Success
Initiating Good Manufacturing Practices (GMP) production operations, whether as part of facility start-up, product changeover, or following a shutdown, requires complex coordination of both logical and physical process controls. Inefficient configuration and qualification practices for automation and information management systems can lead to both start-up delays and productivity losses.
Improve the Flow, then Automate
Flow is the foundation of world class production processes. Impediments to flow result in waste (transportation, inventory, motion, waiting, over-processing, over-production, defects). Unfortunately, digital solutions are often leveraged to address observed wastes in production processes without first addressing foundational problems.
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.