On June 4-7, experts in process analytical technology (PAT) gathered for the 2023 IFPAC conference. While there, they discussed the latest developments in PAT, quality by design, and overall process monitoring and control within the pharmaceutical, biotechnology,...
single use
Holistic Production Readiness Strategy is Key to Operational Success
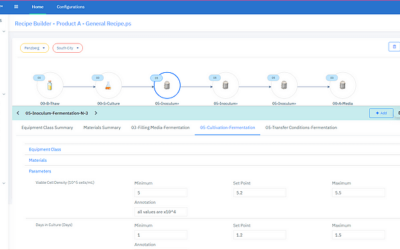
Initiating Good Manufacturing Practices (GMP) production operations, whether as part of facility start-up, product changeover, or following a shutdown, requires complex coordination of both logical and physical process controls. Inefficient configuration and qualification practices for automation and information management systems can lead to both start-up delays and productivity losses.
Single-Use Biopharma Needs Specialized pH Sensing Technology
Industry is seeing a shift to single-use biopharmaceutical manufacturing methods, prompting a need for new pH measurement approaches to overcome instrumentation challenges. Monitoring and historizing the pH of a process media batch, and often using this value for...
Single-Use Pressure Transmitter Receives Pharma Innovation Award
The Rosemount 550PT Pressure Transmitter was recently recognized by Pharma Manufacturing for filling the need for measurement technology designed specifically for single-use biomanufacturing.
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.