I caught up with Emerson’s Chris Amstutz, whom you may recall from earlier Life Sciences-related posts. Chris leads a team of Life Sciences industry consultants. He shared how calibration is a time-consuming and documentation intensive activity in highly regulated industries.
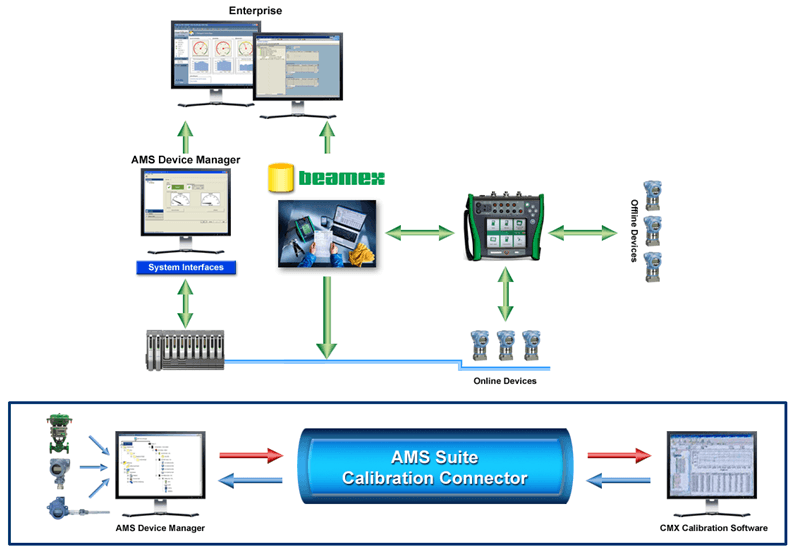
He also pointed me to a relatively new Calibration Excellence whitepaper, which describes a solution to help pharmaceutical and biotech manufacturers reduce their calibration maintenance costs and satisfy the requirements of regulatory agencies. The whitepaper outlines the joint Emerson/Beamex calibration solution that is based upon AMS Device Manager, AMS Suite Calibration Connector, and Beamex CMX software.
The whitepaper defines calibration:
Instrument calibrations involve the comparison of the instrument’s value to a known reference value.
The calibration process is one of the major maintenance activities in terms of spent time:
The Emerson/Beamex calibration solution standardizes the physical calibration procedure with a documenting calibrator so that the documentation and historical records of all calibration procedures are united and automated. This means:Calibrations can take one to four hours per device using generally accepted procedures. Since many plants have thousands of field instruments, a significant portion of maintenance time must be spent on calibration alone. The primary reason for calibration is that even the best measuring instruments or their sensors are not permanently stable. Over time, they can drift and lose the ability to meet their accuracy specifications. Drift, whatever the cause, makes recalibration necessary.
…users do not have to spend significant time manually entering device information for smart assets, such as HART or FOUNDATION fieldbus devices. Since all of this information is already found in AMS Device Manager, the AMS Suite Calibration Connector simply transfers it into CMX, with no manual entry necessary… By using a documenting calibrator, results are automatically stored in the calibrator’s memory during the calibration process. Then the results are automatically transferred from the calibrator’s memory to the computer/database. This eliminates the user step of manually recording the results, making the entire process much faster, less costly, and more accurate.
The whitepaper highlights how the components are interconnected to achieve this automatic capture and flow of information. It also highlights results achieved at a pharmaceutical manufacturer’s site:
GlaxoSmithKline in Cork, Ireland, piloted the Calibration Excellence solution and gained numerous benefits. Don Brady, an automation engineer at GSK, reported that the Calibration Excellence solution helped the facility:
- Reduce the steps in its overall calibration process from 17 steps to 10 steps;
- Remove two steps that each posed risks of human data-entry errors;
- Conduct Emerson and Beamex consulting workshops to review existing processes and optimize and refine the new processes to use improved technologies;
- Use the new system’s flexibility to manage workflow processes.
More on GSK’s calibration process improvements is highlighted in a ControlGlobal.com article, Search for the Asset Management Holy Grail – Part 2.