In a recent post, Collaborating for End-to-End Supply Chain Improvements, we highlighted a Life Sciences symposium where challenges to business improvements were discussed as inputs for technology and solution innovation to address these challenges.
One of the sessions focused on the needs for process intelligence and analytics for pharmaceutical and biopharmaceutical manufacturers. The topic team included leaders from the Life Sciences industry, Emerson local business partners, and Emerson. Emerson’s Jonathan Lustri coordinated this session with an IT Director of one of the leading pharmaceutical manufacturers at the symposium.The focus for these discussions were the application of manufacturing IT within Clinical and Commercial manufacturing and how to realize value from manufacturing IT investments. The team sought to identify common challenges and best practices for system architectures, project implementation and IT systems management.
A common challenge is the performance of the end-to-end supply chain and how can data and information improve its efficiency. The scope of the supply chain includes receiving raw materials to the delivery of medicines to the patient with focus on increasing capacity utilization and reducing cost, product release times and time to market. Also, this data and information needs to be used to improve quality, assure compliance, and increase process understanding.
For manufacturers, cross functional teams including manufacturing, IT and automation professionals need to understand how to use date to impact supply chain operational improvements.
From a manufacturing IT perspective, the scope includes:
- Batch Automation
- Electronic Batch Records
- Multivariate Data Modeling
- Production Management & Scheduling
- Process Analytical Technology
- Statistical Quality Control (SQC); Statistical Process Control (SPC)
- Overall Equipment Efficiency (OEE)
- Laboratory Information Management
- Process History
- Knowledge management systems (big data, search….)
- Training management systems
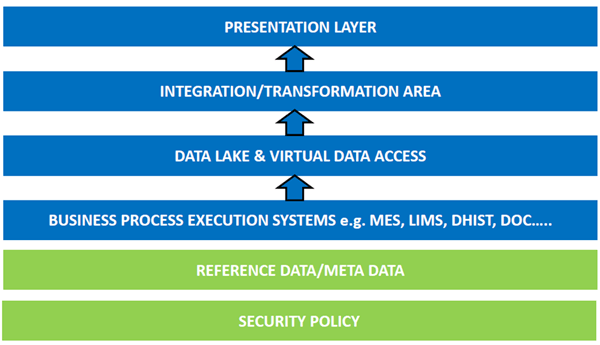
The architecture for these domains runs from the security policy through the presentation layer of information:
During the course of the discussions, numerous common challenges were identified. I’ll highlight just a few. One of the significant ones was seeking better ways to realize value. It often takes more than technology—it takes an organizational cultural change to switch to a data-driven, decision-making mindset versus the traditional risk avoidance mindset common to highly regulated industries.
Another common challenge was the technology transfer through the drug development process from product design (PD) through cGMP full-scale production
Deliverables from these in-depth discussions included documenting these common challenges, developing guidance, best practices and case studies through the pooling of knowledge, all recommendations for standards and technology road maps. All of these deliverables were focused around reducing effort to implement these manufacturing IT systems, technology transfer and product changeover times and focusing activities with team skill sets in mind.
We’ll look at some of the other topic areas addressed during the symposium in future posts.
You can connect and interact with other experts in the pharmaceutical and biopharmaceutical industries in the Life Sciences group in the Emerson Exchange 365 community.