For pharmaceutical and biotech manufacturers, Continued Process Verification is:
…the collection and analysis of end-to-end production components and processes data to ensure process is running under the state of control and product outputs are within predetermined quality limits.
In an earlier post, Online Multivariate Data Analysis for Continued Process Verification, we mentioned a new approach for performing online multivariate data analysis (MVDA) process monitoring that can be used for CPV. This approach involves:
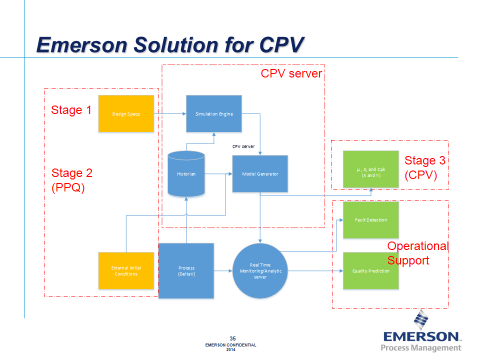
One of the inventors of this new approach, Emerson’s Zuwei Jin, shared some details with me. MDVA can be a powerful extension to CPV. The simulations provide the number of batch histories required to develop the models for MDVA and allow CPV program to be built using MVDA tool as early as from stage 2.…a CPV server that includes an historian, a simulation engine based on random or pseudo-random calculation such as Monte Carlo, and a model builder that incorporate Multi-variate principal component analysis (PCA) and partial least squares (PLS) correlation. The simulation engine also includes a quality predictor with interface for input customer design space and process parameter distribution characteristics.
Typically today, most CPV analysis is performed with trend charts, which measures the quality attribute or process parameter over the number of batches performed. These statistical process control (SPC) charts are univariate and done after the batch completes. Offline correlation may be performed on these quality attributes and process parameters and the impact of discrepancies may be assessed. Looking at individual parameters, such as with the Golden Batch technique, doesn’t take into account interactions between attributes and parameters. Multi-variate data analysis can however identify the outliers even if none of the individual variables deviate from their upper and lower control limits.
By using MDVA, both Enumerative (X and Y) and Analytical (Y->X) statistics are evaluated, it helps not only to see the deviations but also to focus on the cause of deviations and real time monitoring supporting operation decisions becomes possible.
With the CPV server, online MVDA is now relevant during stage 2 of process validation and allows design space to be input to CPV program in the drug development process. The CPV server is built on top of on-line MVDA platform such as DeltaV Batch Analytics (BA) and can provide a seamless transition for process monitoring from clinical manufacturing to commercial production by progressively replacing the simulated batches with real batches for MVDA model building to achieve the ultimate goal of continued process verification (CPV).
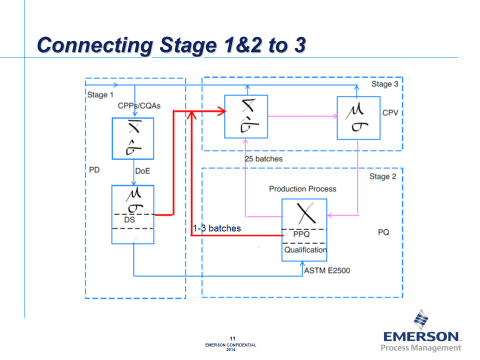
Online MDVA helps to detect process fault and predicts batch quality ensuring process stays in design space during the production process. While providing traditional univariate statistics for CPV monitoring, the CPV server enables problems to be discovered and corrected to also support operational decisions. In addition to allowing to connecting stage 1, 2, and 3 in drug development cycle, Zuwei also noted that the CPV server is well suited for handling large and complicated sets of data in plant wide applications and will make CPV a relative easy practice. He predicts that on-line MVDA will become a common tool in process monitoring and will be a key component of future CPV programs.
You can connect and interact with other pharmaceutical and biotech experts in the Life Sciences group in the Emerson Exchange 365 community.