In this age of rapid races for vaccines and therapeutics to battle the Coronavirus, there is even greater focus on the technology transfer process through the product development pipeline to commercial production. Emerson’s Michalle Adkins and Ron Rossbach will host a webinar on reducing time to market for new therapies—a key opportunity for pharmaceutical and biopharmaceutical manufacturers. This webinar will take place on August 13 at 15:00 CET – Europe / 09:00 EDT.
Michalle and Ron will highlight new developments in Process Knowledge Management, information exchange standards and overall Life Sciences data management and data exchanges that are making this historic “wish list item” for process automation a reality. As common execution system building blocks align with high level process design and development tools, the ability to automate data collection and deploy execution system recipes across the enterprise is expanding.
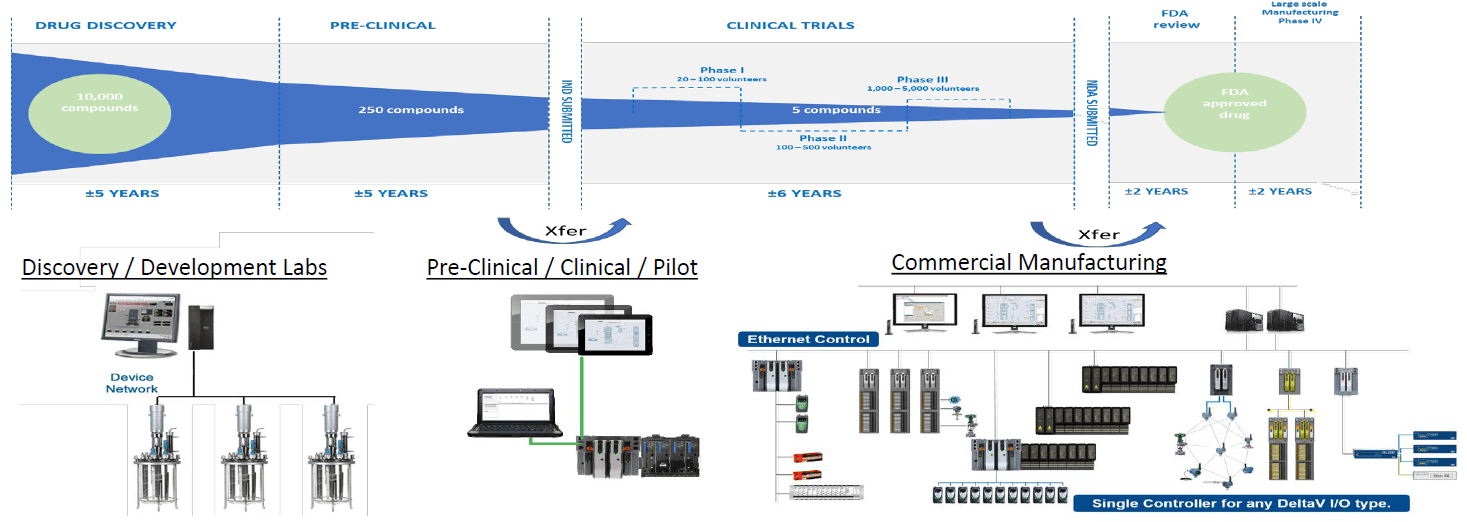
Today the average product development cycle takes 97 months from clinical trials to approval. Transferring data between phases can be difficult, with lost or inaccessible prior stage information. The technology transfer process often requires extensive staff and manual efforts.
Technology can play an important role in consolidating and integrating data across the lifecycle. An important element is to modularize process building blocks. Also, having flexible product and recipe designs can accelerate the product development lifecycle and provide more seamless technology transfer through the journey from basic research through development pilot plant, clinical trials produced from the pilot plant to commercial scale manufacturing.
Automation technology that can scale from the discovery/development labs up to the pre-clinical/clinical/pilot manufacturing and then to full commercial manufacturing simplifies the information transfer across phases in the lifecycle.
Michalle and Ron will discuss other key technologies in the pipeline acceleration process including product demand planning, manufacturing execution, change control, laboratory testing, recipe management, analytics and key production event and data collection.
Register for the webinar for more on how there are significant opportunities to accelerate the development pipeline and overcoming challenges in streamlining work processes for improved business performance. Visit the DeltaV System Life Sciences Solutions section on Emerson.com for more on the technologies and solutions to drive improved operational performance.