Life sciences may be the ideal industry for the implementation of AI technologies. After all, bringing a treatment all the way through the drug development process is an extremely complicated undertaking, with tremendous amounts of data and a wide array of...
MES
Manufacturing Readiness Is the Next Frontier in Life Sciences AI
AI is changing drug discovery faster than most people expected. Commercial manufacturing is next. The organizations best positioned for that shift are the ones preparing now, not because they’re behind, but because the window to prepare thoughtfully is exactly when...
Digital Frameworks Transform Drug Development
In the wake of the COVID-19 pandemic and the associated development of a vaccine in record time, the biopharmaceutical industry has seen significant change. Today, we know that faster development is possible, though it is still challenging. That has increased the...
Part 2: Execution Intelligence: Embedding AI Inside Validated Manufacturing
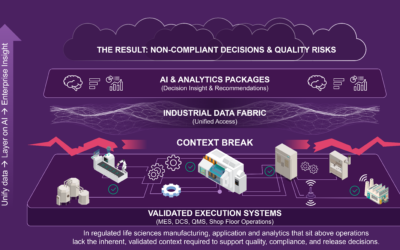
In Part 1, we explored why digital strategies that sit above operations break down in regulated life sciences manufacturing. When analytics are disconnected from validated execution, insight loses the context required to support quality, compliance, and release...
Part 1: Why AI in Life Sciences Must Be Grounded in Validated Execution
The common digital strategy playbook is: unify the data, layer on AI, and drive enterprise-level insight. The strategy sounds compelling in its simplicity. And for some industries and some use cases, it can work. But in regulated life sciences manufacturing, there is...
Bringing Born-Digital Speed to Every Life Sciences Workflow
In the last few years, life sciences innovation has jumped by leaps and bounds. Many of the emerging autologous, patient-specific therapies being released are accomplishing things the general public never thought would be possible. These autologous treatments are...
Preparing Pharma Operations for an Autonomous Future
Continuous manufacturing was a game-changer for the ability to deliver life-improving and life-saving biopharmaceutical treatments to patients as quickly as possible. Automation made it possible to make manufacturing significantly more efficient, helping teams to...
The Road to Self-Driving Labs Starts with Smarter Automation
If there’s one thing that has been repeatedly proven true in the life sciences industry, it’s that automation drives success. After all, automation leads to repeatability, fewer mistakes, higher safety, and faster outcomes. This high success rate for automation in...
The Golden Age of Life Sciences Innovation Needs Smarter Data
There exists an interesting tension in the life sciences industry; while people have access to more treatments for more maladies than at any other time in human history, there is also increasing pressure to deliver new, innovative treatments. For evidence, simply look...
Seamless Automation for a Personalized Future
It’s hard to discuss the life sciences today without talking about advanced therapeutics and personalized medicine. We are currently in a period of massive innovation that will reshape the delivery of medicine for decades to come. Where once pharmaceutical companies...
MES Done Right
Automation in the process manufacturing industry is nearly always complex. Not only must automation technologies carefully orchestrate operations, but they must also monitor safety, reliability, efficiency, and these days, even sustainability. The distributed control...
Life Sciences and Automation Companies Deliver the Future—Together
In the life sciences, bringing treatments to market quicker is no longer a bonus. Today, accelerating the treatment development pipeline is an expectation—arguably a core value—of the world’s most respected pharmaceutical companies. The pipeline for the COVID vaccine...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.