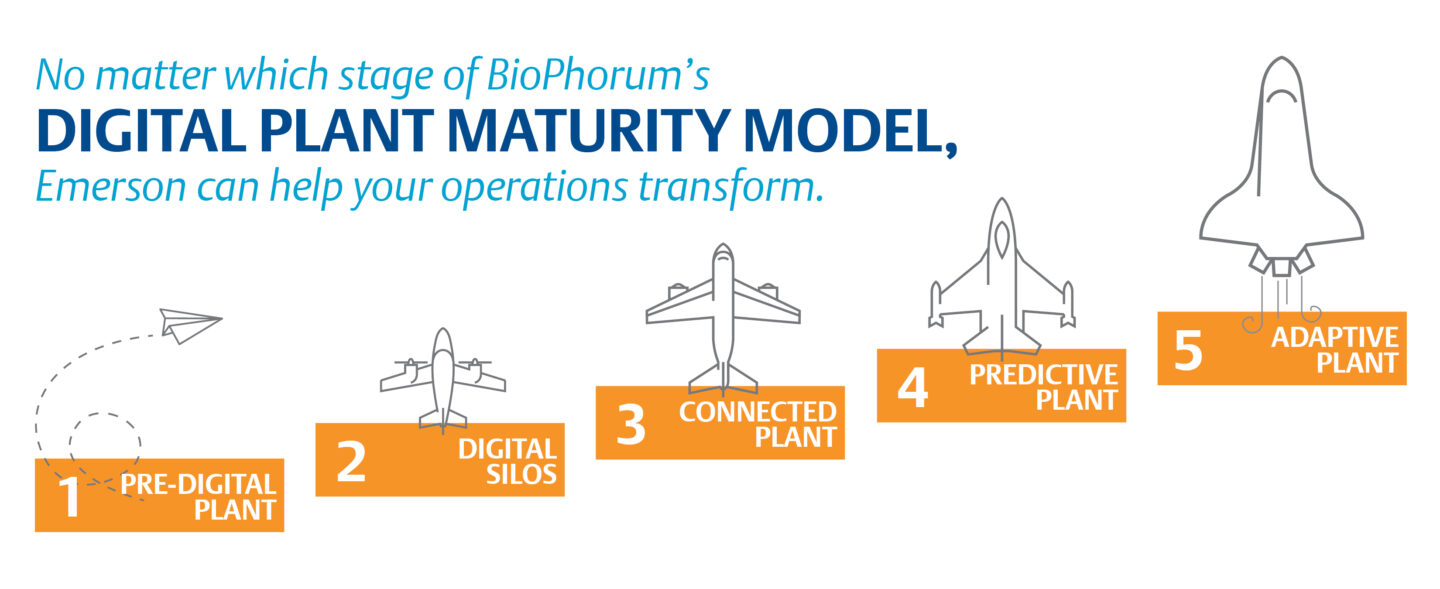
On Monday at the Emerson Exchange Users’ Conference, Emerson’s Michalle Adkins and AbbVie’s Jeff Ziolkowski moderated a panel with Fujifilm’s Soren Skerris, Takeda’s Dr. Nina Schwalb, Eli Lilly’s James Weber, and Emerson’s Christian Berg. The experts shared the strategies they have employed and the lessons they’ve learned on their journey across the BioPhorum Digital Plant Maturity Model toward creating adaptive life sciences plants.
Adaptive acceleration
Driving pipeline acceleration to get critical treatments into the hands of patients faster is more important than it has ever been. The panelists explained how moving toward an adaptive plant is an important step in accelerating the development pipeline, sharing some essential best practices they learned in their own journeys.
One key idea all the panelists shared is that an adaptive plant isn’t an endpoint, but rather part of a journey—and that journey is undergirded by the people who make it possible. James shared that the challenge of building an adaptive plant is
“10% technical and 90% people.”
Nina elaborated, explaining that education and transparency are paramount. Generating buy-in is critical to changing the skillsets of the people the plant depends upon to implement new strategies. The technology, Chrisitan explained, is only the means to an end that empowers the people who will make the real change happen.
Avoiding the roadblocks
The panelists also explored the barriers inherent in moving to a more adaptive manufacturing model. Once again, the plant’s most valuable resource—its personnel—were at the heart of the issue.

Soren explained that in some cases, a new process might not be easier or better for the people who are operating it but may still be significantly better for the overall development and manufacturing scheme. Successfully implementing such changes requires transparency to help people understand the overarching vision. And just as importantly, James shared, the stakeholders need to know they have an organizational structure that works and has a clear vision—a vision, Nina explained, that must lay out clear goals and responsibilities.
Modularity is key
The panelists also shared other critical elements of building an adaptive plant, including the importance of flexibility and modularization. Not only do flexibility and modularization enable a plant to pivot when strategies and needs change, but it also simplifies validation. Soren offered an example,
“We predefine specific capabilities that we call ‘process actions’ and those process actions are built in a way that you can put them together for whatever sequence of operations you want. That way, you don’t have to validate the whole process every time.”
Nina elaborated,
“We validate the use case, but we don’t re-validate everything that’s enabling it. That’s the key for us, and we do it in a cookie-cutter style globally.”
There are many ways to start the journey toward more adaptive life sciences manufacturing, but, as the panelists explained, the concept of the journey is key. The BioPhorum Digital Plant Maturity Model is a great place to start that journey, helping you both build a roadmap for your digital transformation journey and quantify your current capabilities and understand the challenges you may face.
There’s still a lot more to come from this year’s Exchange! Stay tuned to the Emerson Automation Experts blog for regular updates throughout the week! And if you’re at the conference, I hope to get the chance to meet with you and hear some of your experiences!






