How many of you have more passwords than you can possibly remember? Count me in with the affirmatives. Biometric authentication is one way to overcome this large and growing problem. For process manufacturers and producers, especially those in highly regulated...
Operations & Business Management
Automating Tank Farm Systems and Work Practices
Have you ever stopped to consider the path and ownership transfers your car's gasoline or diesel took from the wellhead when it was still crude oil until you pumped it into your tank at the gas station? For the businesses involved in custody transfer process through...
Deploying Syncade Manufacturing Execution System in a Non-English Language
Author: Jonathan Lustri I recently worked on a Syncade manufacturing execution system (MES) project where the system was deployed in Danish. Emerson executes all its projects with a global team and this one was no different. Our customer was Danish and we had team...
Niche-Buster Medicine Information Solutions
Author: Michalle Adkins We are seeing a trend in the life sciences industry moving away from blockbusters to niche busters, population based medicines and even to the extreme of personalized medicines. The Emerson Life Sciences consulting team recently had the...
Managing Coalbed Methane Wells Feeding LNG Plant
Liquefied natural gas (LNG) facilities require careful pressure management on their incoming gas pipelines. In a recent LNG Industry article, Turn it Up (and Down), Emerson's Michael Calvert describes the challenges and solutions around the Santos Gladstone LNG (GLNG)...
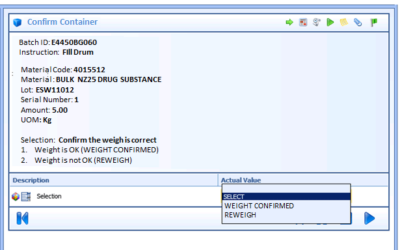
Optimizing Batch Process Automation Design in MES and DCS
For pharmaceutical and biotech manufacturers, the flow and capture of information is as important as the flow of the manufacturing process in readying products to release for sale. In a Pharmaceutical Technology magazine article, Connecting MES to Process Control,...
US FDA Quality Metrics Guidelines Update
Author: Eric Kuebler Since the Continued Process Verification (CPV) guidance in 2011, the U.S. Food and Drug Administration (FDA) has been talking quality and verifying process state of control. Now the FDA is getting around to defining what that looks like and what...
Conference Room Pilots to Generate Involvement in MES Projects
Author: Jonathan Lustri As a Life Sciences consultant, I get involved in many different aspects of MES [manufacturing execution system] projects. In a recent project, I helped define the project execution plan. I knew Emerson's biotech customer was highly concerned...
Reducing Communications Problems during Shift Changeovers
Sources of unplanned downtime can be many including equipment failures and human error. For downtime resulting from operator interactions, one major source of errors is the communications breakdown that sometimes happens during a shift change. If not told or seen in...
Prototype Manufacturing Operations Management Projects
The ANSI/ISA-95 standard defines models for the information flows between the instrumentation and automation layers up to the enterprise information layers for process manufacturers and producers. Moving up the levels increases the need for people, processes and...
Continued Process Verification in the Process Validation Lifecycle
In 2011, the U.S. Food and Drug Administration (FDA) issued a Guidance for Industry – Process Validation: General Principles and Practices. It highlighted a third validation stage goal of continued process verification (CPV) for: …continual assurance that the process...
RFID Tagging for Materials Movements
Some of the technologies making the Internet of Things (IoT) possible include WirelessHART, radio-frequency identification (RFID), near field communication, barcodes, QR codes and digital watermarking. I mention this because many process operations require material...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.