How many of you have more passwords than you can possibly remember? Count me in with the affirmatives. Biometric authentication is one way to overcome this large and growing problem. For process manufacturers and producers, especially those in highly regulated industries such as pharmaceutical and biopharmaceutical manufacturing, authenticated actions also need to be recorded to prove that the manufacturing process was done in compliance with what was designed and validated.
I caught up with Emerson’s Jay Jeffreys on what manufacturers were doing to simplify the authentication and recordkeeping process in these regulated industries to meet legal and compliance requirements.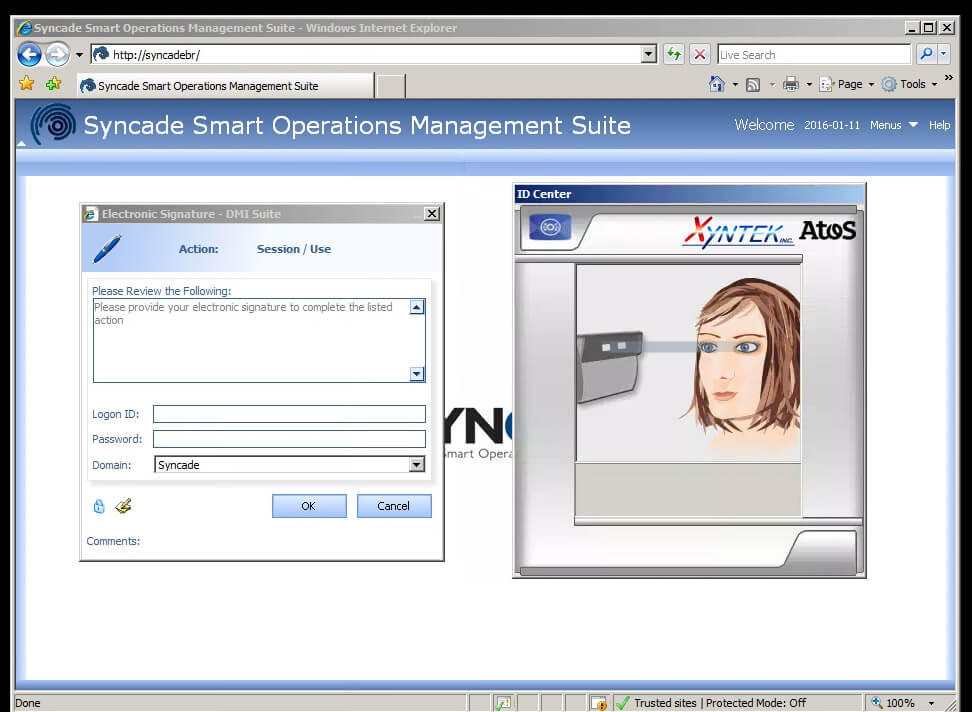 He described how the Syncade Operations Management software integrates Xyntek’s biometrics solutions, which compares the physical user biometric scan against stored biometrics data. Some forms of identification may include a person’s iris, fingerprint, palm-vein or voice. Once the authentication occurs, this confirmation is added to the electronic records managed by the Syncade software of the product being produced.
He described how the Syncade Operations Management software integrates Xyntek’s biometrics solutions, which compares the physical user biometric scan against stored biometrics data. Some forms of identification may include a person’s iris, fingerprint, palm-vein or voice. Once the authentication occurs, this confirmation is added to the electronic records managed by the Syncade software of the product being produced.
Beyond minimizing security risks associated with lost/stolen passwords and identification cards, this biometric authentication method can speed up signoff times and provide documentation that is more complete. This process helps shifts the focus from paperwork to maintaining the quality of the products produced. Jay noted that operational savings of 50% or more could be realized, primarily in areas of data capture, document preparation, and review.
For regulatory requirements such as the U.S. Food and Drug Administration’s 21 CFR Part 11 compliance for system security, user access, and electronic signatures is a matter of who you are versus what you know and what you have. Manufacturers that have installed biometrics have realized cost savings by replacing typical username and password entries with quick and simple scans, reducing the level of IT support required.
Moving the biometric authentication process into the electronic records provides improved searchability, easier-to-prove compliance for signing and witnessing steps and a shorter cycle from end of batch production to ready for sale with the complete electronic record produced in parallel with the batch.
Integrating biometric authentication into electronic records is not limited to highly regulated industries but any industry where transactions and workflow steps require proof of action by individuals. For more, visit the Syncade Operations Management Suite web pages.
You can also connect and interact with other operations management and manufacturing execution experts in the Operations Management group in the Emerson Exchange 365 community.

