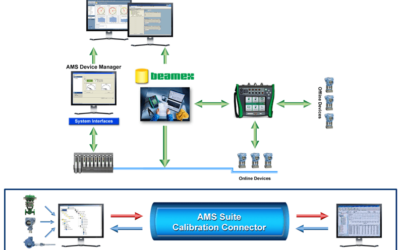
I caught up with Emerson's Chris Amstutz, whom you may recall from earlier Life Sciences-related posts. Chris leads a team of Life Sciences industry consultants. He shared how calibration is a time-consuming and documentation intensive activity in highly regulated...
life sciences
Avoiding Moisture Variability in Fluidized Bed Dryer Operations
In an earlier post, Trends in Process Analytical Technology and Quality by Design, I highlight a point made in the video, "…PAT methods around product moisture, which projected a $2 million annual savings in improved product quality." The methodology used for the...
Trends in Process Analytical Technology and Quality by Design
At the recent conference for pharmaceutical and biotech manufacturers, Interphex 2012, Pharmaceutical Manufacturing magazine Editor in Chief Agnes Shanley discussed trends in the industry's use of process analytical technology (PAT) and quality by design (QbD) with...
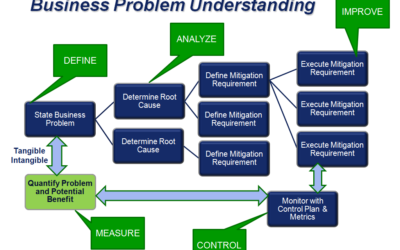
Using Operational Excellence Techniques to Understand and Solve Business Problems
Pharmaceutical and biotech manufacturers, like other process manufacturers, are under constant pressure to improve the performance of their business—product quality, safety, plant efficiency, and throughput. Emerson's Michalle Adkins, a senior consultant on the Life...
Managing the Pharmaceutical and Biotech Technology Transfer Process
In a Research and Markets report, Technology Transfer Strategies - A Guide to Maximizing Returns Within the Pharmaceutical and Biopharmaceutical Industries, the authors note: The efficient and effective transfer of new technologies is widely recognized as being a key...
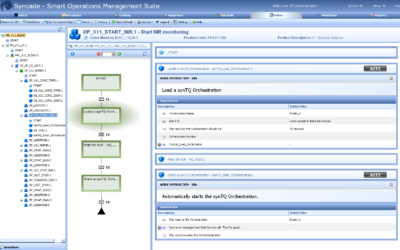
Faster Product Releases with PAT Methods in Production Recipes
This week, the International Foundation Process Analytical Chemistry (IFPAC) is holding their 26th international forum and exhibition in Baltimore, Maryland USA. At this event, Emerson's Chris Amstutz, Life Sciences industry consulting team director, gave a...
Continuous Process Verification per FDA Process Validation Guidance
It was just about a year ago that the U.S. Food and Drug Administration published their Guidance for Industry - Process Validation: General Principles and Practices. I caught up with Emerson's Heather Schwalje, a senior consultant on the Life Sciences industry team....
Shrinking that Cycle Time
An Emerson fellow blogger, Alan Babbitt, and his Emerson Global Life Sciences Blog was first to the scene to cover a great Bioprocess International article, Hurry Up and Wait? The article, by Emerson senior Life Sciences industry consultant, Michalle Adkins, describes...
Critical Backup for Your ISA95 Level 3 Information
I caught up the other day with Emerson's Shenling Yang, who is a senior project execution engineer in the Life Sciences and Food & Beverage (LSFB) industry group. You may recall Shenling from earlier posts. She shared two pieces of news with me. The first is that...
Applying a Structured Methodology to PAT Initiatives
In a recent Pharmaceutical Processing magazine article, PAT Searches for its Identity, author Bikash Chatterjee discusses the seemingly slow pace of Process Analytical Technology (PAT) implementations. The article states: What the FDA has provided is a bold chance for...
Five Strategies for Mitigating Automation Project Risk
Every capital project has it challenges, usually around tight budgets and tight schedules, not to mention team flexibility and changes in scope. Recently at the Interphex conference, the big industry gathering for Life Science manufacturers and suppliers, Emerson's...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.