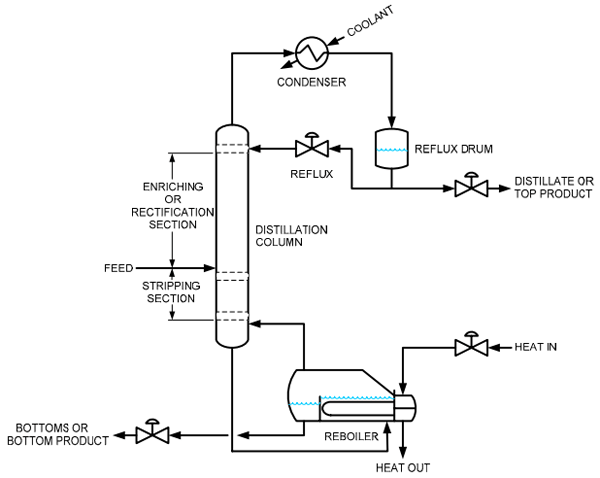
In part 1, we looked at how control has been traditionally performed on distillation columns. We’ll look at some improvements in today’s post.
Inferential Composition Measurement
One thing required for distillation column control is product quality measurement. This is typically done with analyzers, but many columns are not equipped with online analyzers. Columns equipped with online analyzers often have extended sample times to get product quality results.
However, product quality is related to temperature and pressure. The relationship is strict for a binary mixture and often “good enough” for a mixture with more than 2 components. Temperature control in a distillation column is really inferential control as the temperature is used to infer purity. There is an argument to be made for using pressure compensated temperatures, especially if pressure varies much.
Antoine’s equation is used to uniquely relate pressure and temperature for a pure component in vapor-liquid equilibrium as a linear function of the log of pressure and the inverse of temperature. For a mixture of 2 components, purity can be inferred from the Antoine relationship by considering the slope, which will vary with composition.
If there is a pressure swing, the temperature will change immediately according to the ideal gas law. But eventually the vapor-liquid equilibrium will shift and the pressure and temperature will rebalance as the purity changes.
Control of pressure compensated temperatures, if done right, can help improve composition control. I have had great success with pressure-compensated temperatures, especially in light hydrocarbon service. Other inferential model forms can be used as well, perhaps including multiple temperatures measured in different locations in the column.
Basic regression models are often used, and I have had success using Artificial Neural Network models in the DeltaV system. Still, there will always be a need for an analyzer to ensure product purity is in spec, even if it is done off-line. An inferential measurement gives the control system something to control between analyses, whether they are performed online or offline in a lab.
Control Design
While single loop control is common in distillation, there has been a lot written about more complex controls to operate closer to product specs and process constraints. If feed varies, simply ratioing the main streams to feed can be helpful. Product (both distillate and bottoms) to feed, reflux to feed, and even steam to feed ratios can minimize the disruption when feed changes.
How should you pair the control loops and why? F Greg Shinskey and others did a lot of work on distillation control and applied the concept of relative gain array analysis (RGAA) to control of distillation. Usually it is good enough to control the top composition with reflux and the bottom composition with reboil and close the material balance with the product flows on level control.
But at times it would be better to pair differently. This is because of the loop interactions mentioned previously. The RGAA approach tries to identify the best pairings for the most stability. One of the things that fell out of the Shinskey work was the notion of using reflux to distillate ratio to control composition. The reflux ratio (or another similarly derived relationship) is a measure of the amount of heat used per unit amount of material processed, which is closely related to product purity.
The reflux ratio control works in part on the assumption that level is well controlled. If the level varies greatly and especially rapidly, then the reflux ratio isn’t a true measure of reflux to product because of the change in accumulation.
In part 3 of the series, I will look at how to close the material balance and how advanced process control tools such as model predictive control can help in operating the distillation columns closer to constraints.
From Jim: You can connect and interact with other advanced control experts in the Control & Safety Systems groups in the Emerson Exchange 365 community.