At Texas A&M University’s Mary Kay O’Connor Process Safety Center’s 76th Instrumentation and Automation Symposium, Emerson’s Greg McMillan presented two topics. I’ll recap his Bioreactor Control and Optimization presentation in today’s post and save his Process Effects on Control for a future post.
Greg opened explaining how digital twin technology enables Process Analytical Technology (PAT) for manufacturers in the Life Sciences industry.
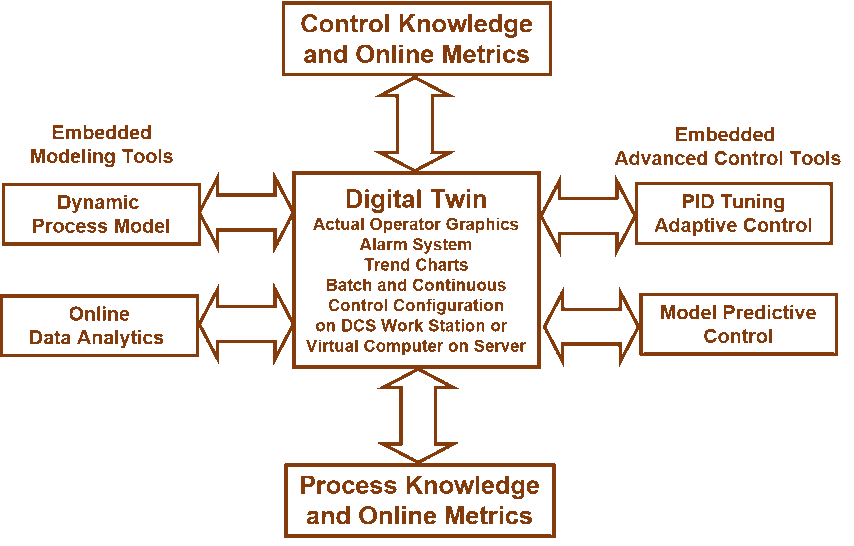
He described some of the automation dynamics blocks that enable the digital twin in PAT—Analyzer, Backlash Stiction, Sampler, PID Performance, and Future Value. Analyzer simulates at-line and offline analyzer variable cycle time and analysis time with sensitivity and resolution limits. Backlash Stiction simulates valve deadband, resolution. lags, and delays (all aspects of ANSI/ISA 75.25.01 – Test Procedure for Control Valve Response Measurement from Step Inputs). Sampler simulates digital device response including wireless transmitter trigger level, default update rate and trigger update rate. PID Performance provides online loop metrics for load and setpoint response: integrated error, peak error, overshoot, undershoot, settling time and rise time. Future Value provides a fast calculation with good signal to noise ratio of future value and rate of change to prevent the U.S. Environmental Protection Agency’s RCRA [Resource Conservation and Recovery Act] pH violation and surge plus give online identification of surge curve and optimization of batch end point and setpoint response.
Greg showed Texas A&M’s digital twin with a bioreactor model and advanced process control operating at 500 times real-time speed.
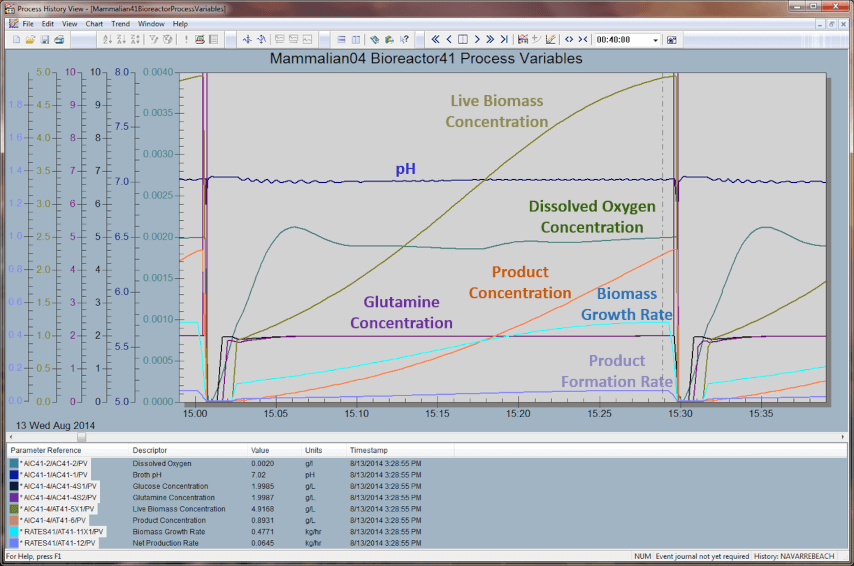
He explained the numerous components in the model including growth and formulation rates, Michaelis-Menten Kinetics, and growth rates vs. dissolved oxygen, glucose, lactate, pH and temperature. Greg also shared some considerations in enabling the model speedup including that it’s the product of the kinetic and media speedups, making sure the integration step is less thank ¼ continuous volume residence time, multiplying valve & pump capacities, flow measurement, and flow controller by the kinetic speedup factor, and several other considerations.
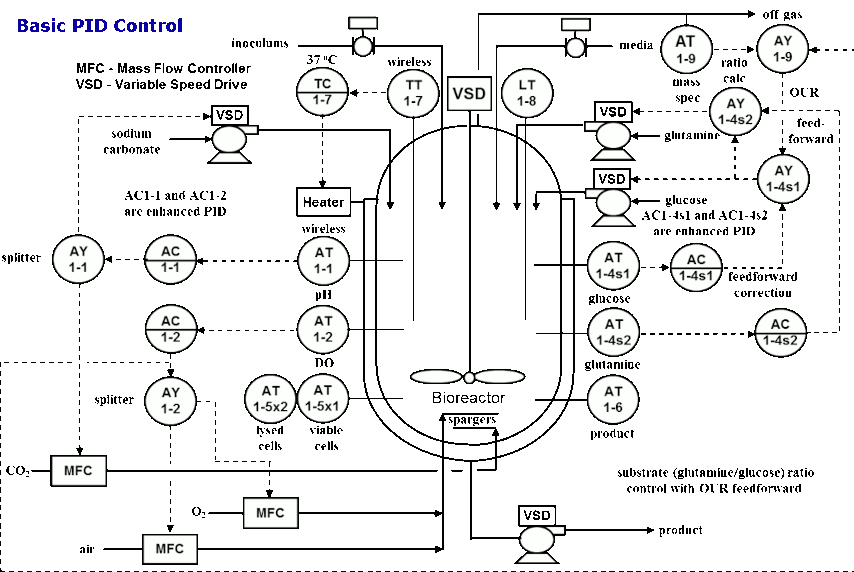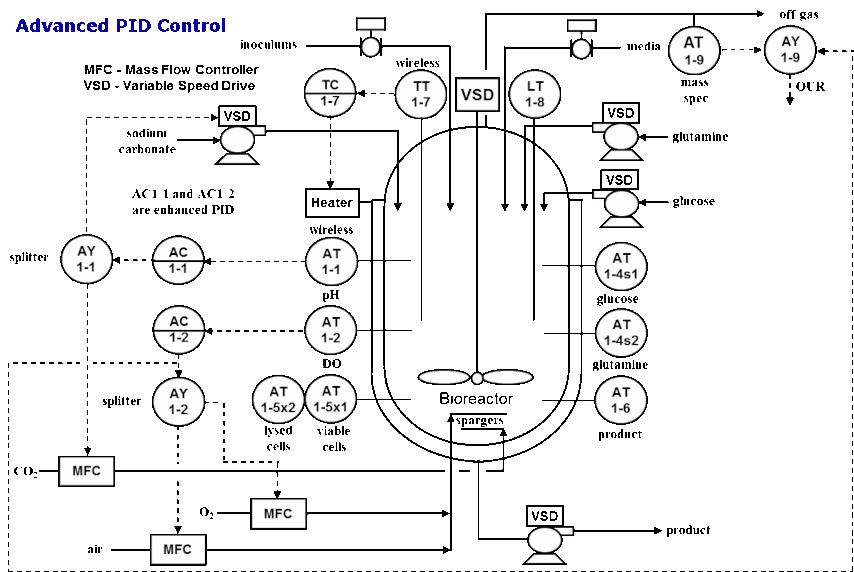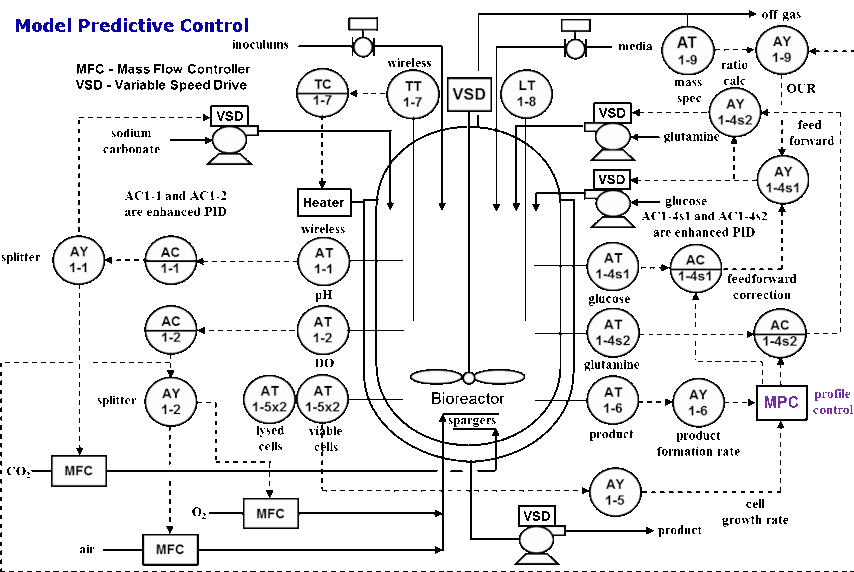
He also shared the control strategies for basic PID control, advanced PID control and model predictive control (MPC).
For deeper insights into bioprocessing modeling and control, order Greg and his fellow authors’ book, New Directions in Bioprocess Modeling and Control. Greg notes that this book provides practical, comprehensive knowledge on how to use the advances in analytical measurements, and basic and advanced control to provide greatly improved batch profiles and endpoint consistency. It reveals breakthroughs that enable dynamic models to be readily parameterized from a plant’s data historian trend charts. The consequential integration of the measurements, models, and controls into a digital twin with recently developed blocks to provide profiles and predict endpoints enables nonintrusive experimentation at several hundred times real time. The resulting improvements in batch cycle time and consistency can be designed, tested, quantified, and confirmed and operators can be trained independently of actual plant operation. The benefit from the elimination of a bad batch — particularly for new biologics — is potentially worth ten or more million dollars.





