
Most people have heard the adage that, “COVID changed everything.” However, few people, when presented with that idea, think that any of those changes were positive. But we often forget that in the span of eight short months, the world saw a wide array of innovators and manufacturers come together to deliver a vaccine—a process that typically takes upwards of 15 years.
In his recent article in Industry Week magazine, Emerson’s Nathan Pettus explores the impact this rapid treatment development is having on the world. Nathan explains,
“Life sciences manufacturers have learned many lessons on the value of this acceleration, and it tantalizes the imagination for what could be.”
One click technology transfer
“What could be” is actually tremendously exciting. Biopharma organizations, now aware of what can be done, are taking the steps to ensure it will be done. One of the areas of the development pipeline where this new move toward acceleration is most obvious is in technology transfer, the formalized process for moving manufacture of a treatment from one facility, scale, or development pipeline phase to another.
Traditional technology transfer is extremely complex and time consuming, mainly because the process today is largely manual. Nathan explains,
“To transition a product between stages, someone must translate what the people in the previous stage were doing so they can then make processes work on the next scale of equipment, usually via trial and error, potentially adding years to the development pipeline.”
But a new solution focused on digitalizing the process into a centralized transport and translation platform—one click technology transfer (OCTT)—aims to change all that. OCTT will create a framework to seamlessly interconnect the many disparate systems teams across the development pipeline use:
- Enterprise Resource Planning
- Quality Management
- Automation Systems
- Computerized Maintenance Management Systems
- Process and Knowledge Management
These, and other critical systems, will be integrated into a single, cohesive ecosystem. When OCTT reaches maturity, every team across the development pipeline should be able to easily move critical data forward and backward across research and development and manufacturing to eliminate the biggest delays in drug treatment design and manufacture.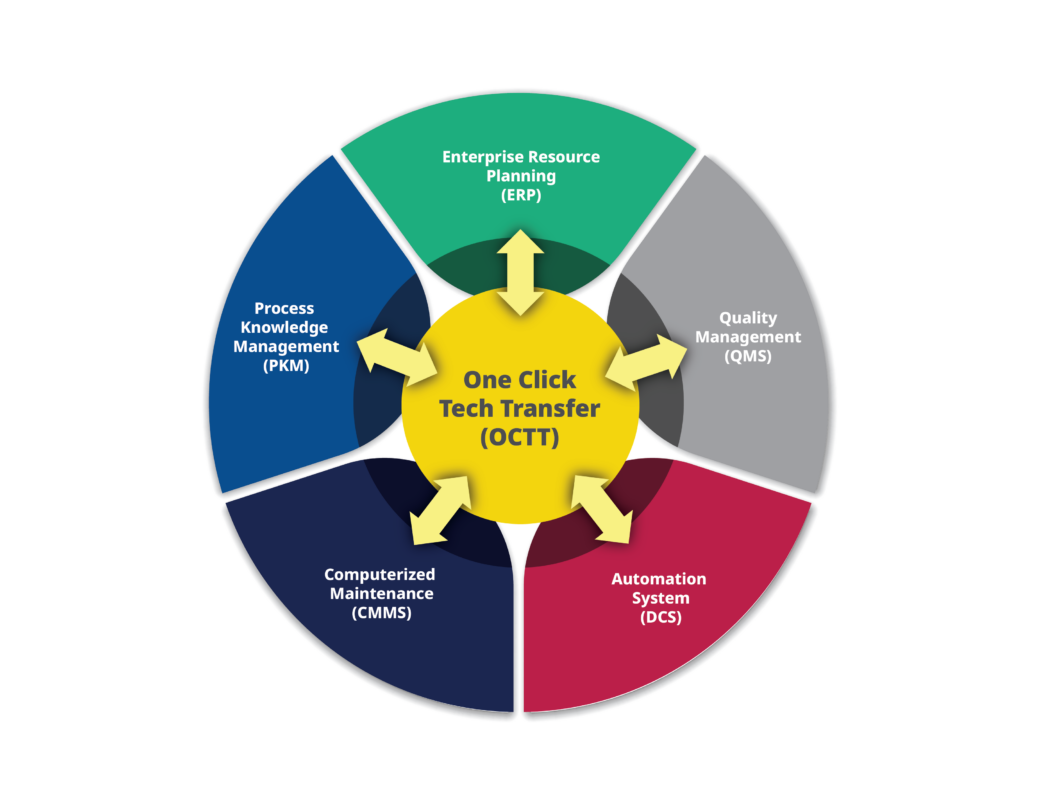
The first steps
The earliest stages of development of the OCTT platform are already underway. Tying systems together to build a workable OCTT framework will require open standards and commonality in goals. To meet this need, Emerson is establishing a grassroots executive board of key life sciences stakeholders to begin developing standards, as well as creating an OCTT Center of Excellence for research and development of new technologies. Together, the executive board and Center of Excellence will help steer the implementation of the best standards and most powerful technologies.
It’s unclear how long it will take until technology transfer is truly “one click.” However, what we do know is that the concentrated movement in that direction by Emerson and its partners on the executive board will begin establishing technologies that will reshape the way we look at the life sciences development pipeline. And as that acceleration dovetails with new treatment methods like personalized medicine, the sky is the limit for the future of biopharma. It’s an exciting road ahead.

