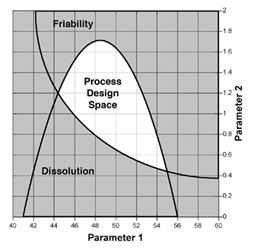
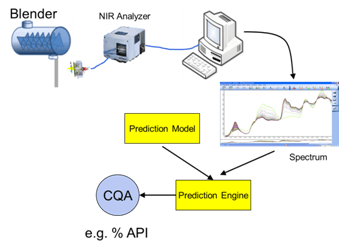
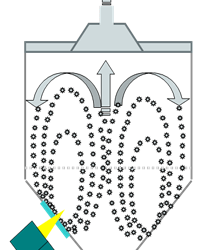
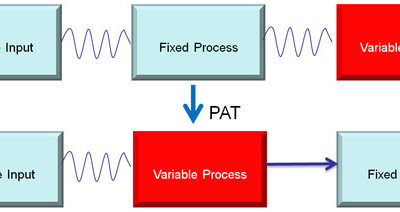
For pharmaceutical and biopharmaceutical manufacturers, there are a lot confusion around the concepts of design space (DS), Process Analytical Technology (PAT), and Quality by Design (QbD). Emerson's Zuwei Jin believes that this confusion has largely limited the...
Process Analytical Technology
Enabling Release by Exception Manufacturing
The Emerson Exchange conference October 6-10 in Orlando, Florida USA is rapidly approaching. I caught up with Emerson's Michalle Adkins who mentioned that her team of Life Sciences consultants would be highlighting the enablement of release by exception. Although...
Implementing Process Analytical Technology and Continuous Process Verification
In the 4:42 video, Life Science Drug Process Development and Manufacturing, Emerson's Gary Mitchell highlights the changes occurring for pharmaceutical and biotech manufacturers. Gary opens noting how these manufacturers are challenged to respond to new methods for...
What Do You Do for a Living?
Author: Michalle Adkins I often get asked, "So, what do you do for a living?" Boy is that a loaded question. When I say that I manage a group of Life Sciences Industry Consultants for Emerson—that really does not help to answer the question. Hmmm, how do I describe...
Implementing Quality by Design
Many manufacturers in the Life Sciences industry are challenged to respond to a new paradigm for process development and manufacturing. The FDA's cGMP for the 21st Century initiative is driving the industry to change its development and manufacturing to be based on...
Continuous Manufacturing for Solid Dose Form Pharmaceuticals
Emerson's Jonathan Lustri explores the trend towards continuous manufacturing for solid dose pharmaceuticals and the technologies and practices that make this possible. This past year I have become aware of a very big change that is coming to the pharmaceutical...
Fluidized Bed and Bin Blender PAT Applications
In the early 2000s, a Wall Street Journal article, New Prescription For Drug Makers: Update the Plants, shared the news: The FDA [U.S. Food and Drug Administration] has concluded that the industry needs to adopt manufacturing innovations, partly to raise quality...
Establishing a Process Analytical Technology Program
We're now just a couple of weeks away from the September 30-October 4 Emerson Exchange conference in the Dallas/Fort Worth area of Texas. Today's the last day to save $200 on the conference fee, so register today if you're planning on joining us. If you're in the...
You Already Asked Me That!
I recently saw an interesting discussion about the use of consultants. One question that came up was why they seem to ask the same question in slightly different ways or the same question to different functional groups in an organization. Emerson's Michalle Adkins, a...
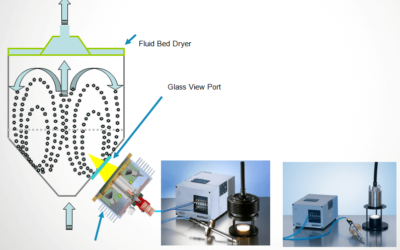
Avoiding Moisture Variability in Fluidized Bed Dryer Operations
In an earlier post, Trends in Process Analytical Technology and Quality by Design, I highlight a point made in the video, "…PAT methods around product moisture, which projected a $2 million annual savings in improved product quality." The methodology used for the...
Trends in Process Analytical Technology and Quality by Design
At the recent conference for pharmaceutical and biotech manufacturers, Interphex 2012, Pharmaceutical Manufacturing magazine Editor in Chief Agnes Shanley discussed trends in the industry's use of process analytical technology (PAT) and quality by design (QbD) with...
Interphex 2012 – Real-Time Release in Production Recipes
I noted in yesterday's post that two major industry conferences were happing this week—the pharmaceutical and biotech industry's Interphex show and the oil and gas industry's Offshore Technology Conference (OTC). I highlighted happenings from OTC in that post. Today...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.