We’re now just a couple of weeks away from the September 30-October 4 Emerson Exchange conference in the Dallas/Fort Worth area of Texas. Today’s the last day to save $200 on the conference fee, so register today if you’re planning on joining us.
If you’re in the pharmaceutical and biotech manufacturing industries, you may want to catch the workshop, 8-4621 – Establishing a PAT (Process Analytical Technology) Program.
 It is scheduled for Tuesday October 1 at 3pm and repeated on Wednesday at 9am. Emerson’s Gary Mitchell is teaming up with a biotechnology manufacturer on this workshop. Gary has frequented the Emerson Exchange Americas on a number of occasions in his previous life as an Emerson “user” for a biotech manufacturer, where he was the PAT Program Manager.
It is scheduled for Tuesday October 1 at 3pm and repeated on Wednesday at 9am. Emerson’s Gary Mitchell is teaming up with a biotechnology manufacturer on this workshop. Gary has frequented the Emerson Exchange Americas on a number of occasions in his previous life as an Emerson “user” for a biotech manufacturer, where he was the PAT Program Manager.
Their workshop:
…will cover a real life example of establishing a PAT Program within a large biotech company, covering the following:
- Team Structure, Team Charter
- Governance and buy-in from Senior Management
- PAT as part of QBD initiative
- Initial Projects – low hanging fruit
- Moving from Development into Manufacturing
- Program current status
- Next steps, including Emerson Batch Analytics & SynTQ
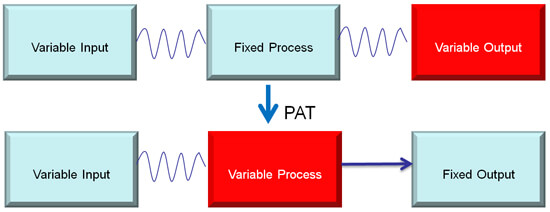
I have a sneak preview of the presentation. Gary opens with background on Process Analytical Technology noting that it’s about process automation. It converts the age old “fixed process” to “variable process” by increasing process understanding, using analytics, instruments, and process control, and focusing on attributes versus parameters.
The objective is to attain Quality by Design (QbD) with the production process by implementing technologies that enhance process understanding and better control the manufacturing process. PAT projects should solve current technical problems, support innovation, and improve efficiency in development, manufacturing, and quality assurance. The benefits should be clear in meeting business needs, increasing process control and understanding, reducing operating costs, reducing risk, and improving success rates.
PAT encompasses concepts including “design space”, process understanding, continuous process trending, multivariate analysis, improved process control, and process analytical technologies. Design space is the improved understanding and management of product and process variability. This understanding leads to earlier recognition of atypical process performance, streamlined process validation, continuous process improvement, and greater flexibility to meet regulatory requirements.
Gary highlights the importance of established PAT processes and documentation including the PAT Charter, which contains the strategy and plan and the PAT projects listing with their priorities and status. Budgets across departments should be coordinated and information sharing sites such as SharePoint should be established to make sure lines of communication are clear.
Gary and Joe close the workshop sharing some workflows to facilitate problem solving and implementation of new technologies. They highlight perspectives on transforming data into information, recent accomplishments at the biotech manufacturer’s facility and business results achieved.
It’s a presentation you’ll want to schedule for lessons on applying PAT in your pharmaceutical or biotech manufacturing process. Also, you’ll want to schedule Emerson’s David Rehbein‘s EDU-5367 – Batch Analytics educational course on how to use DeltaV Batch Analytics Viewer to monitor performance of a batch using real-time and historical data. He’ll interact with session attendees to show how this application assists in predicting product quality, detect process faults, and provide reasons for deviation so that operators can take action in real time.