In the early 2000s, a Wall Street Journal article, New Prescription For Drug Makers: Update the Plants, shared the news:
The FDA [U.S. Food and Drug Administration] has concluded that the industry needs to adopt manufacturing innovations, partly to raise quality standards. The agency is overhauling its elaborate manufacturing regulations for the first time in 25 years.
 So over the past decade, the Life Sciences industry has been incorporating these changes using a framework known as Process Analytical Technology (PAT). Emerson’s David Rehbein shared the presentation podium at the recent Emerson Exchange conference with a staff member of a leading pharmaceutical manufacturer to share some PAT solutions that have been implemented. The two applications they shared included a fluidized bed dryer and a blender.
So over the past decade, the Life Sciences industry has been incorporating these changes using a framework known as Process Analytical Technology (PAT). Emerson’s David Rehbein shared the presentation podium at the recent Emerson Exchange conference with a staff member of a leading pharmaceutical manufacturer to share some PAT solutions that have been implemented. The two applications they shared included a fluidized bed dryer and a blender.
They opened by describing how PAT is defined:
PAT is considered to be a system for designing, analysing, and controlling manufacturing through timely measurements of critical quality attributes and performance attributes…….. with the goal of ensuring final product quality.
As we described in an earlier post, Establishing a Process Analytical Technology Program, PAT helps to convert a “fixed process” to a “variable process” by increasing process understanding, using analytics, instruments, and process control, and focusing on attributes versus parameters.
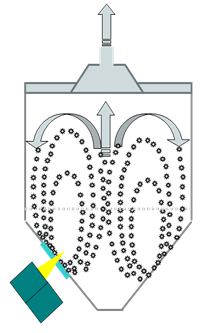 For the fluidized bed application, one issue was that overdrying could result in flaking tablets being produced. Underdrying would cause the produced powder to stick to the punching tools. Also, extra cycle time was incurred to re-dry the produced powder after the initial endpoint of the batch. This variability in moisture content negatively impacted overall capacity utilization, energy costs, and labor costs.
For the fluidized bed application, one issue was that overdrying could result in flaking tablets being produced. Underdrying would cause the produced powder to stick to the punching tools. Also, extra cycle time was incurred to re-dry the produced powder after the initial endpoint of the batch. This variability in moisture content negatively impacted overall capacity utilization, energy costs, and labor costs.
The pharmaceutical manufacturer worked with the Emerson Life Sciences consulting team to perform a study to test a proposed solution involving a Bruker MATRIX-F FT-NIR process spectrometer. The team gathered data to refine the moisture prediction model during the test period and used it in the implementation of the solution for fluidized bed moisture control.
The solution involved the NIR probe performing contactless measurements through a window into the fluidized bed. Also, the solution included data coming from a PLC/HMI control system to a PAT server running synTQ client/server software and a second instrument workstation running a synTQ instrument server and Bruker OPUS spectroscopy software.
The results of these real-time quality measurements around moisture were reduced operator labor, reduced energy consumption, reduced product retesting, and the potential for elimination of manual moisture testing.
 For the NIR (near-infrared) spectroscopy blend monitoring application, content uniformity is the major critical quality attribute (CQA) for the mixing and drying of the powered material prior to compression and tableting. Before the PAT solution was applied, blending operations were based on pre-determined time intervals or a fixed number of blender revolutions. This method was highly dependent on operators to monitor the process through visual inspections and off-line testing.
For the NIR (near-infrared) spectroscopy blend monitoring application, content uniformity is the major critical quality attribute (CQA) for the mixing and drying of the powered material prior to compression and tableting. Before the PAT solution was applied, blending operations were based on pre-determined time intervals or a fixed number of blender revolutions. This method was highly dependent on operators to monitor the process through visual inspections and off-line testing.
The PAT solution involved a bin blender controlled by a PLC with a Brimrose NIR spectrometer collecting spectral data of the blend contents. The data fed an instrument PC running synTQ and Brimrose Snap32! spectrum analytical software. The results of from this analysis were communicated back to the PLC by the synTQ FM software running Orchestrations (PAT methods).
What happens is that an operator first executes a spectrometer suitability test. Next they trigger the synTQ Orchestration for NIR blend control where the Orchestration auto-starts the blender via OPC communications. The operator observes the computer display for end of blend. When synTQ determines the end of blend condition, it issues a remote stop command to the blender. The operator then executes another spectrometer suitability test.
The end result of adding real-time quality monitoring when fully implemented at scale is expected to be better blend uniformity using a fully validated PAT execution method, reduced cycle times for the blending operation, continuous improvement through KPI measures, 21 CFR Part 11 compliance, and a path to real-time release in the future.
