Sean Buckley and co-authors from Roche, Takeda, and BioPhorum author CGT [cell & gene therapy] Actors and Process Maps which highlight six common types of CGT showing how the pattern of actors and process blocks varies by therapy type.
Sean Buckley
BioPhorum Cell and Gene Therapy Personas and User Stories
The BioPhorum CGT Personas and User Stories toolkit details the needs of all the key players involved in end-to-end cell and gene therapy (CGT) processes. It can be used by anyone who wishes to better understand how IT systems can support the manufacture and delivery of CGTs.
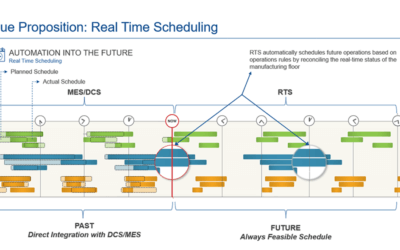
BioPhorum MES of the Future
In BioPhorum’s MES of the Future Manifesto, Sean and Kate Porter collaborated with other biomanufacturing leaders on requirements for future Manufacturing Execution Systems.
Avoiding Unplanned Downtime in Life Sciences Industry
For manufacturers in the Life Sciences industry, production challenges often occur in these four areas—production readiness, equipment reliability, personnel effectiveness and facility/line efficiency. In this post, I’ll focus on equipment reliability, and specifically, unplanned downtime.
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
The opinions expressed here are the personal opinions of the authors. Content published here is not read or approved by Emerson before it is posted and does not necessarily represent the views and opinions of Emerson.