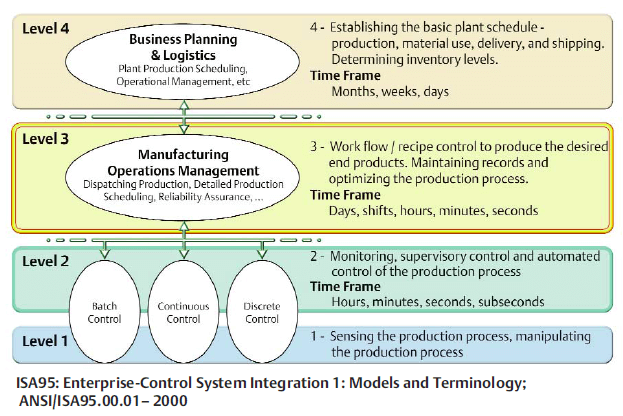
Projects to connect your process operations to your business processes and workflows typically require connections with several specialized software platforms. For manufacturers in the pharmaceutical and biotech industries, these electronic connections span the ISA-95 Enterprise-Control System (IEC 62264) hierarchy.
 I caught up with Emerson’s Gary Mitchell, a member of the Life Sciences industry consulting team. Gary highlighted four major areas where these connections occur as well as typical platforms found in these areas.
I caught up with Emerson’s Gary Mitchell, a member of the Life Sciences industry consulting team. Gary highlighted four major areas where these connections occur as well as typical platforms found in these areas.
At the highest level of the ISA-95 hierarchy are the business systems. These include Enterprise Resource Planning Systems (ERP) such as SAP or Oracle/JD Edwards, Warehouse/Inventory Management Systems (WMS), which are often a part of the ERP, and Production Scheduling Systems (PSS), such as Preactor.
Below the business systems are the Manufacturing Operations Systems, which include process control systems (PCS), data historians, process analysis systems (PAS), and Process Analytical Technology (PAT) systems. The DeltaV system is an example of a PCS, OSI PI a data historian, SIMCA-Batch On-Line (SBOL) as PAS, and SynTQ a PAT.
Quality systems play a key role in the success of an integrated manufacturing enterprise. Gary highlighted four areas including Laboratory Information Management Systems (LIMS), such as Thermo Scientific LIMS, Electronic Document Management Systems (EDMS), such as Documentum, Exception Management Systems (EMS), such as TrackWise, and Training Management Systems (TMS), such as Qumas.
The final area Gary shared with me is Facilities Systems. These include Building Management Systems (BMS), Computerized Maintenance and Management Systems (CMMS), Calibration Management Systems (CMS), and Asset Management Systems (AMS). Popular applications in these four areas respectively include Siemens APOGEE, Blue Mountain, Maximo, and Emerson’s AMS Suite.
In most instances, pharmaceutical and biotech manufacturers do not have a software platform in each of the categories mentioned above. Manufacturing execution software, such as Syncade Suite, can be used to integrate these applications and include capabilities found in several of the categories. For example, modules include Equipment Tracking, Materials Management, Training & Development, and Weigh & Dispense. For optimizing operations, capabilities include Electronic Workflow, Order Management, and Recipe Authoring.
For connecting systems together electronically, Syncade modules include Message Broker, Operations Dashboard, and Process Analyzer. Numerous Quality & Compliance modules can also fill in capabilities that you may not already have in your installed software platforms.
The key in all this integration activity is to understand the sought business objectives, key stakeholders/departments, the current workflows, and the desired future state of the operations. From there, a phased approach can be planned and implemented, designed for early successes and continued efficiency gains. Gary and the Life Science consultants are often involved and can help with these early fact-finding and planning processes.
| iTunes