In an Intech article, Three strategies help life sciences companies implement more successful review by exception, Emerson’s Emilee Cook shares ways for these manufacturers to improve this workflow.
GAMP 5
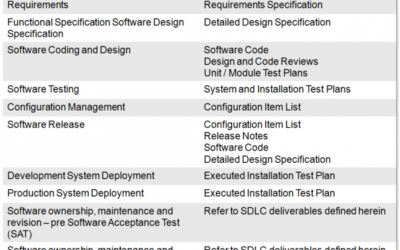
Managing the Software Development Lifecycle for Validated Processes
For those in the biotech and pharmaceutical manufacturing industries, Emerson's Chris Amstutz has been sharing a great series of forum posts in the Emerson Exchange 365 community, Life Sciences track. Here are a few examples of some specific SAP to manufacturing...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
The opinions expressed here are the personal opinions of the authors. Content published here is not read or approved by Emerson before it is posted and does not necessarily represent the views and opinions of Emerson.