Pharmaceutical and biotech manufacturers have stringent regulatory compliance requirements. These requirements must balance with the need for improvement and optimization to run safely, efficiently, profitably and in line with the business and quality objectives.
 In a Pharma Bio World article, The Crossroads of Compliance, Audit Readiness, Profitability, and Data Integrity, Emerson’s Michalle Adkins opens:
In a Pharma Bio World article, The Crossroads of Compliance, Audit Readiness, Profitability, and Data Integrity, Emerson’s Michalle Adkins opens:
True compliance and audit readiness starts with a well thought out, holistic, structured approach that aligns with an organization’s business and quality objectives. Profitably and safely managing a batch production facility while maintaining the compliance and quality of the product and production facility is a challenge that every pharmaceutical and biotech company faces.
Michalle notes that the integrity of the data from the manufacturing process and associated business processes are key to understanding and detecting sources of variability to:
…prevent deviations…essential to running an operationally excellent facility. There are several ways to reduce variability. Mistakeproofing processes, eliminating sources of controllable variability, and knowing how to respond to the remaining sources of variability are all important methods to consider.
Michalle describes several ways to attack variability. Near the top:
…is automating production and business processes. Automation is a solution that enables predictability, detection of variability, and the ability to appropriately respond to remaining variability. Compliance ready systems along with a structured project approach are keys to ensuring the data integrity of these types of solutions.
Involving experienced people is also important, whether an:
…internal resource or an external consultant have a breadth of experience working with many companies in the industry to take advantage of best practices, incorporate a view of where the industry as a whole is heading, and know what automation solutions are available.
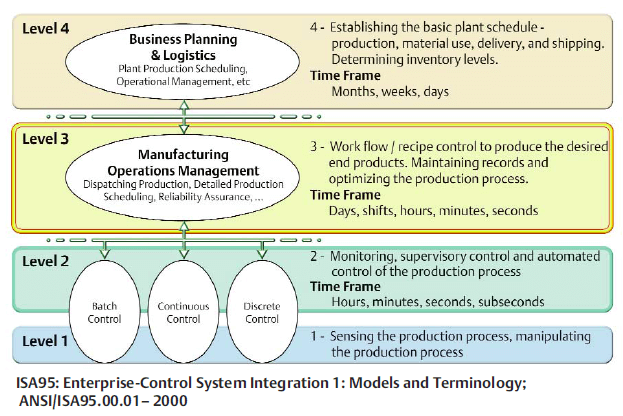
From a technology perspective, Michalle highlights the importance of the ISA-95 model as guidelines for addressing business and manufacturing needs and integrating automation at all levels.
For Layer 1:
…the focus is on properly sensing and manipulating the production process. The primary concern for level 1 is standards adherence: equipment must comply with and enforce quality regulations.
…
It is important to select intelligent sensors that enable prediction of downtime and reduce process variability by keeping devices and instruments performing at their best.
Improving data integrity includes smart instrumentation that is:
…delivered pre-calibrated with documentation and certification to ensure that regulatory standards are met, building compliance into the architecture.
At Layer 2:
…the focus shifts to monitoring processes, providing supervisory and automated control of the production process. With proper automation, organizations can limit variability, which means simplifying compliance.
Batch manufacturing requires sophisticated sequencing across a plant, while at the same time ensuring that only the correct equipment is used, while in the correct state. Defining the process specific requirements and using best in class process control systems (PCS) are keys to success at this layer.
Layer 3:
…connects the concepts of the organizational management system (ERP) to the activity taking place on the production floor.
Michalle cautions:
…operating without a data integrity plan often results in production managers trying to connect isolated islands of process, quality, operations and logistics data by paper, databases, and other mechanisms which may incidentally cause a data integrity issue.
…
An MES [manufacturing execution system] solution can be used to help minimize paper-based traps with electronic verifications of equipment and recipes to reduce variability and deviations, reducing errors associated with these activities. When properly integrated with the PCS and asset and machinery management, the MES provides access at time of use, ensuring all production records can be easily produced, stored, and approved with electronic signatures.
Layer 4:
…is the layer at which business planning and logistics happen. At this level, planners develop the basic plant schedule, monitor material use, track ordering/delivery, and schedule shipping of product.
Business performance is impacted by inventory control and shipping schedules.
If production and materials use records don’t correlate with inventory and delivery records, and organization can find itself producing too little product, or having to store excess inventory that simply sits around.
Read the article to see specific ways that technologies can address the data integrity and work processes at each layer to improve compliance, audit readiness and overall business performance.
You can also connect and interact with other pharmaceutical and biotech experts in the Life Sciences and Operations Management groups in the Emerson Exchange 365 community.