One of the trends in the biopharmaceutical industries that have taken root over the past decade and a half is the use of single-use, disposable equipment in the manufacturing process. Prior to their use, these facilities relied on inflexible, hard-piped equipment. An American Pharmaceutical Review article highlights the benefits of single-use equipment:
I caught up with Emerson’s Zuwei Jin about this trend and how it affects the process control and manufacturing execution strategies. Zuwei explained that single-use equipment is getting more and more popular in Life Sciences industry, particularly for upstream bioreactors and buffer/media preparations. From an economics perspective, total cost of ownership on a commercial scale is better than the traditional stainless steel, fixed installation approach. He notes that single use plus manufacturing execution systems (MES) also seems to be the fastest approach to “proof of concept” of a new drug.Single-use disposable devices and systems have reduced overall contamination rates, enhanced production throughput efficiency resulting in decreased manufacturing operation and maintenance costs. Recent collaborations between leading filtration, container /closure, mixing and fermentation / cell culture suppliers has resulted in fully integrated Single-Use Disposable Bioprocessing Platforms and Systems (SUS.)
The challenge comes from the process control and manufacturing execution side of things. Some of these challenges include the setup of the verification/flowpath confirmation, verification of the equipment process connections from material item scanning, and how the procedural control logic and recipe designs are engineered to name a few.
As Zuwei and some of the DeltaV and Syncade technologists were thinking about these challenges, the idea hit them to develop an interface for single use process verification/qualification instead of the traditional MES procedural control way. This object-oriented interface would be developed to collect parts/lots information of the single-use components and feed back to the MES.
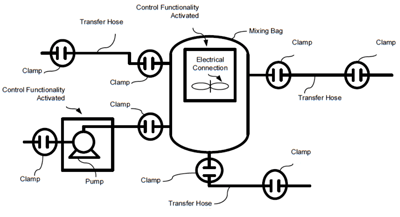 Proximity switches would be used on the clamp/holders connecting the single-use equipment to the process piping. These switches would alert the control and execution strategies that the equipment was connected and ready to be used. Additional integrity tests were included to further assure that these connections were properly secured. The idea of these clamps with proximity switches is that they would have multiple touching points to each side with a digital input back to the control system which might be wired or wireless.
Proximity switches would be used on the clamp/holders connecting the single-use equipment to the process piping. These switches would alert the control and execution strategies that the equipment was connected and ready to be used. Additional integrity tests were included to further assure that these connections were properly secured. The idea of these clamps with proximity switches is that they would have multiple touching points to each side with a digital input back to the control system which might be wired or wireless.
Once all the connections are confirmed this equipment setup information and bill of materials is compared and verified with the material information that is stored and verified in the MES. With the checks completed, the single-use equipment is made active and ready for use in the batch process.
These ideas are another example of the Industrial Internet of Things taking simple items like clamps, which connect single-use equipment to the process piping, making them intelligent, and using this intelligence as part of the control and manufacturing execution strategies to simplify the configuration, ongoing maintenance and improved information for the electronic batch record required to sell the manufactured product.
You can connect and interact with other pharmaceutical and biotech experts in the Life Sciences group in the Emerson Exchange 365 community.

