The Interphex conference for pharmaceutical and biotech manufacturers is underway this week in New York City. Emerson’s Bob Lenich is presenting Big Data in Life Sciences Manufacturing.
Bob opens citing one pharmaceutical manufacturer who generates 5000 data points every second during a production run. This data rate translates into 1.3*1010 pieces of data per production run.
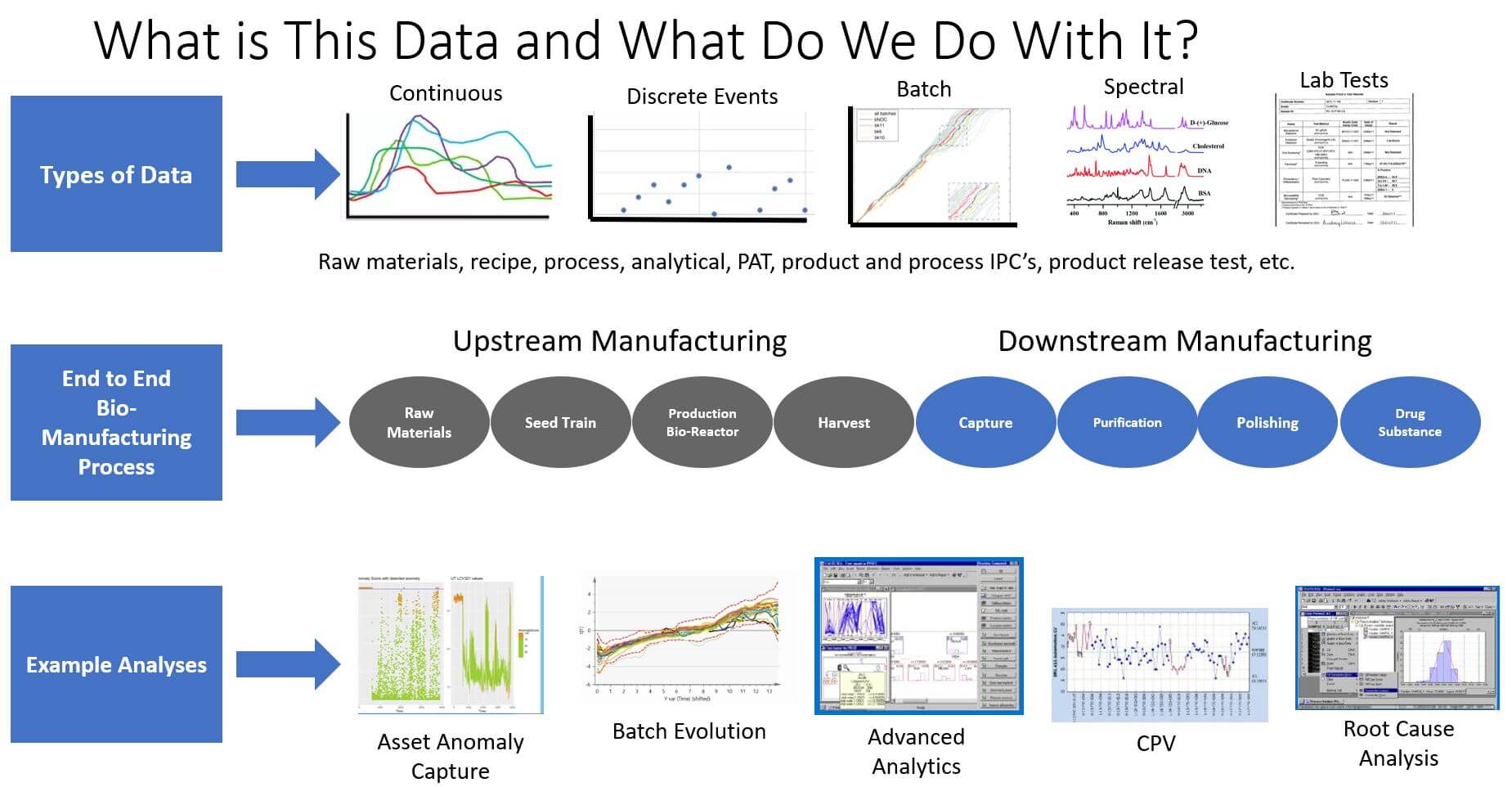
Here are some of the data types and examples across a biomanufacturing process from end to end.
Bob highlights the role of the BioPhorum Operations Group in driving a dynamic and collaborative technology management process with the goal to accelerate innovation and help define future needs, difficult challenges and potential solutions for the industry.
He describes some of the market trends and business drivers that drove the work of Biophorum participants. Market trends include cost pressures from payers, biosimilars & product development, regulatory/demand/competition uncertainty, market growth, and new product classes. To respond to these trends, biopharmaceutical manufacturers are looking to reduce CAPEX and OPEX costs, increase manufacturing flexibility, increase speed from raw materials to final release, and increase quality.
Bob shares some numbers from BioPhorum’s 1st edition technology roadmap. From a speed perspective, in-line monitoring instead of offline lab measurements can reduce the time to release a product from 4-6 weeks to 1-2 days. Projections from the participants project improvements in other areas such as buffer preparation, virus detection, plug and play projects and standard facility designs, and converting large volumes of data into actionable information.
Some big data business use cases where multivariate analysis can be applied include continuous process verification (CPV), advanced process monitoring, advanced process prediction, advanced process optimization, review by exception/real-time release, predictive maintenance, and facility optimization.
He highlights an example of a pharmaceutical manufacturer who identified a pH sensor drift problem two hours before normal testing & reporting normally occurs. This predictive maintenance early notification saved the batch from being scrapped.
Another example is several manufacturers performing batch end-point prediction and soft sensors to move off-line measurements on-line to improve right the first-time processing.
A final example I’ll share from Bob’s presentation is the BioPhorum proof-of-concept testing using a bioreactor as the example skid. Suppliers and end users participating in this testing include Emerson, Rockwell Automation, Siemens, Merck, Pall, and Sartorius Stedim Biotech. The goal is to demonstrate possibility of interactively running a bioreactor phase using more than one control system and more than one equipment supplier’s bioreactor.
If you’re at the Interphex conference, connect with Bob in the Emerson booth #3453. If not, visit the Life Sciences & Medical section on Emerson.com and connect with other experts in the Life Sciences group in the Emerson Exchange 365 community.