One of the key stages delaying and increasing costs in life sciences projects is equipment validation. In fact, it is one of the elements of project completion that project teams are most eager to compress because the value of every incremental improvement is so great. As Michalle Adkins explains in her recent article in Pharma Manufacturing,
“Ultimately, the total cost and time of the full validation process can be five to ten times what was spent on the equipment— a controller that costs $5,000 to purchase can easily cost more than $25,000 to validate.”
But, as Michalle explains in her article, modern software, technology, and methodologies—particularly the control system—are making it easier than ever to compress the equipment validation timeline across all three stages: installation qualification, operational qualification, and performance qualification.
Eliminating mapping streamlines installation
Traditional methods for equipment installation make it difficult and costly to implement the late stage changes that will come when any project goes from engineering and design to implementation. Michalle explains,
“Even when changes are easy to physically implement, the time and costs associated with added documentation and testing to prove correct installation for the change are often prohibitive.”
To avoid the complex mapping that makes late-stage changes more difficult, teams are turning to modern control systems offering the flexibility of I/O on demand. These interchangeable I/O cards connect to the control system via Ethernet and are automatically mapped to the control system for faster installation.
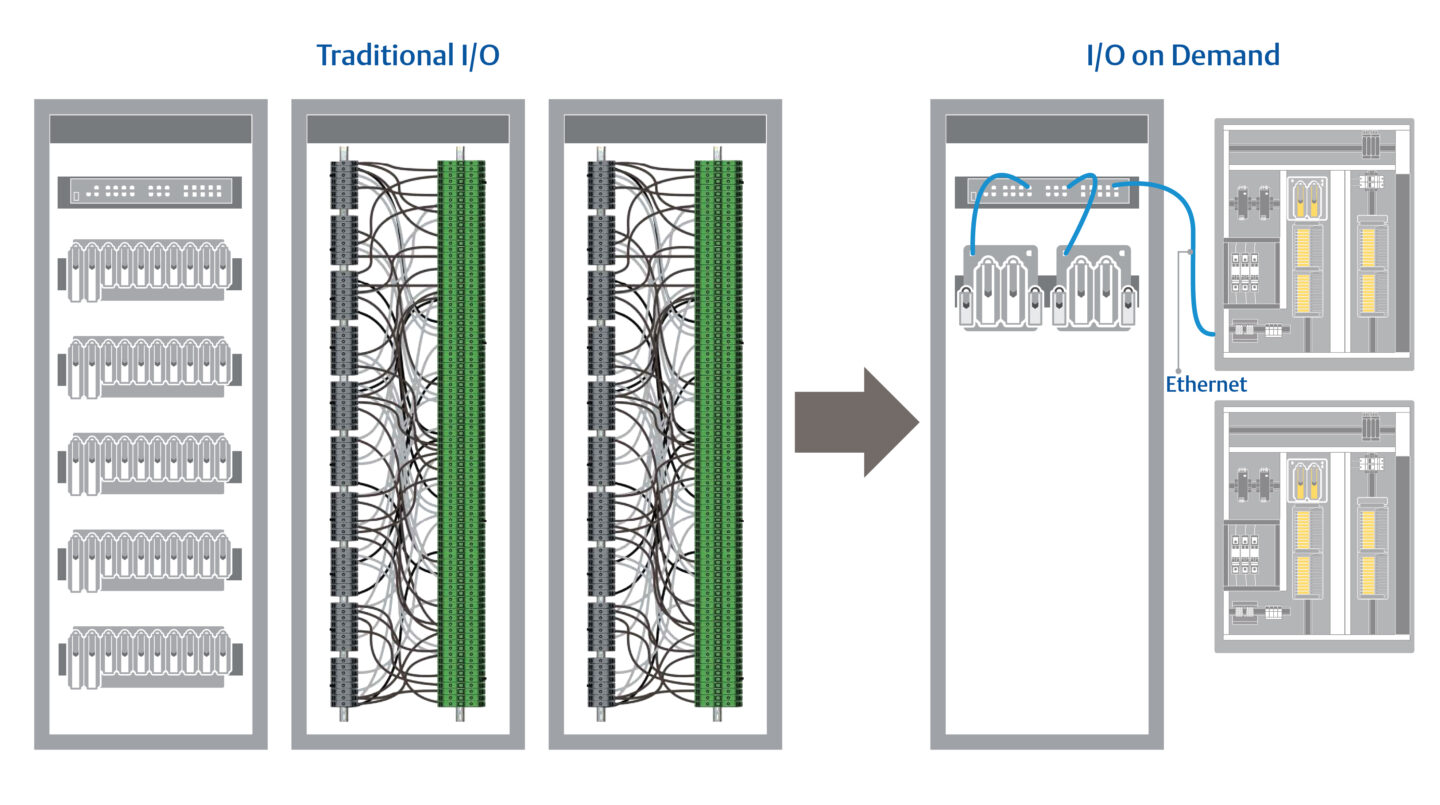
I/O on demand helps eliminate complex wiring and mapping, dramatically shortening validation steps.
New technologies such as Module Type Package (MTP) are also making it easier to integrate equipment into the control architecture. Using MTP, teams can install equipment almost as though it was plug-and-play, eliminating hours of work connecting new machines to the control system.
No need to reinvent the wheel
One of the biggest frustrations in operational qualification is the need to test each unit instance independently regardless of its similarity to other units in the plant. Modern, class-based control systems eliminate this struggle, instead grouping similar units together for faster testing.
“With a class-based system, project teams can test once, after which they only need to test any minor differences among units. The off-the-shelf control system software verifies that everything maps, and the only remaining task is to validate the tags. For systems with duplicate equipment, class-based control can cut the validation time by at least half, and even if there are small differences among units, the project team can map those differences and test only the variations.”
And when teams leverage ASTM E2500 lean validation guidelines, they can compress operational qualification even further. Tools like remote, cloud-based factory acceptance testing eliminates entire stages of configuration to tightly compress timelines.
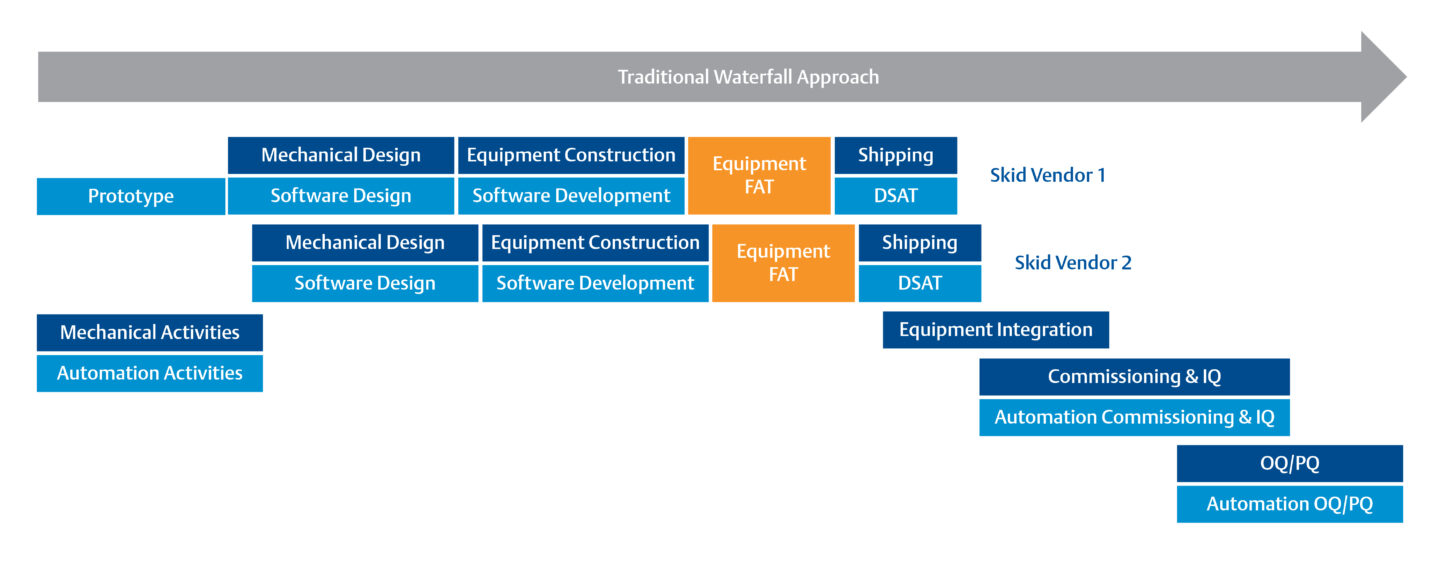
Traditional approaches to operational qualification are time consuming and costly.
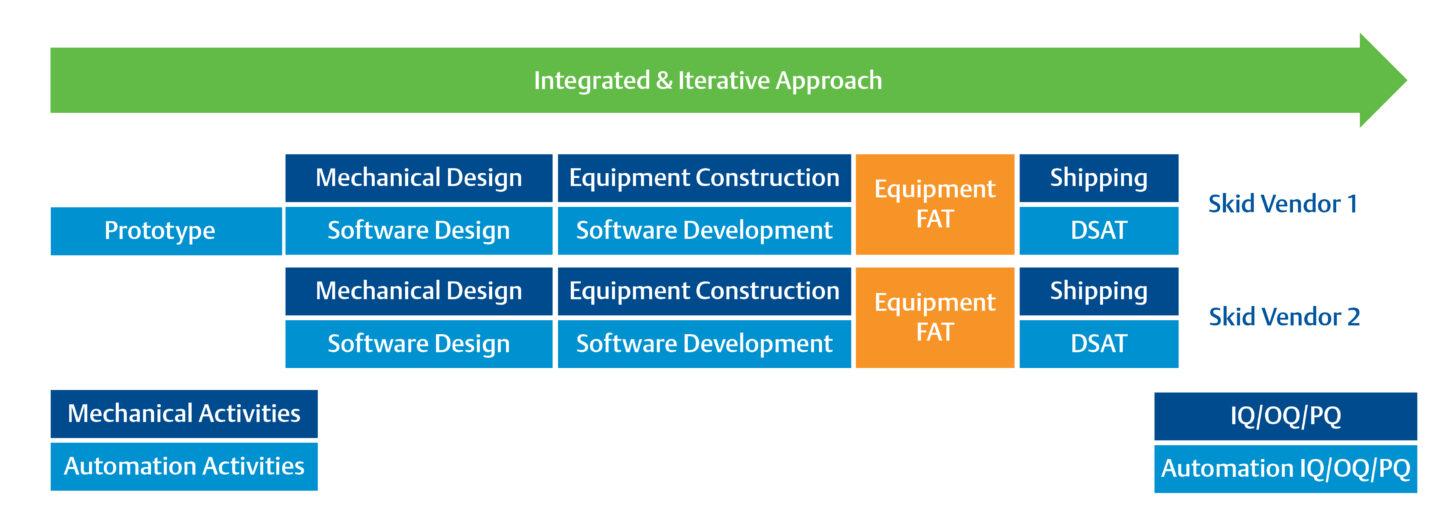
An integrated and iterative approach to operational qualification dramatically reduces project timelines.
Don’t get caught up in the final stages
The goal of performance qualification is to prove that the plant’s processes run properly. But much of the data necessary to demonstrate compliance is often stored in siloed systems. Michalle explains,
“If process, cleaning, and sterilization data are all stored in separate siloed systems, the validation team must spend time finding that data, standardizing it into a usable format, and manually documenting performance qualification test results. Each of these steps takes time, and each is subject to human error that might result in missed data or inaccurate transcription.”
Modern control systems eliminate the time spent transferring and transcribing critical data from a range of siloed systems. Instead, data is centralized in the control system for fast collection, where qualification teams can run reports to rapidly compile all the data they need.
The system at the heart of projects and performance
The first step in streamlining equipment validation should be evaluating whether your current control system helps or hinders your process. Modern control systems like Emerson’s DeltaV™ distributed control system are designed to help project teams ensure equipment is installed correctly the first time and that all processes perform to specifications. You can learn more about how using a modern control system in conjunction with lean manufacturing methodologies can help shorten your equipment validation timeline by reading Michalle’s article in full at Pharma Manufacturing.
I’d also be interested to hear what strategies your team employs to shorten validation and improve speed to market. Feel free to comment below with your thoughts!
