Traditional manufacturing practices have served the biopharmaceutical industry well for years, but times are changing and a need for speed to market in life sciences while reducing manufacturing costs is putting a focus on enabling real-time release (RTR). Technologies such as advanced process control (APC), predictive analytics, in-line monitoring (ILM), and real-time release testing (RTRT) are critical to the new strategies that will bring treatments to patients faster – both faster to market with new products and a faster release for existing products.
Emerson’s Michalle Adkins recently worked with a BioPhorum team of industry experts to author a paper detailing how adoption of ILM and RTR can improve biopharmaceutical manufacturing, providing benefits in efficiency and yield and reducing time to market.

The pitfalls of today’s release strategy
Fast technology transfer gets a lot of attention these days. As manufacturers turn to Process Knowledge Management products like Fluxa, they are dramatically reducing the time spent in tech transfer; however, if they manage to increase that speed only to have finalized product waiting for release testing, much of the valuable speed is quickly forfeited.
Much of today’s release testing is performed offline, after production is complete. The process can occur several weeks after production, causing significant delays and compounding lead time for critical products.
“[Product release] includes reviewing in-process control data, batch records, test records, and off-line release testing of drug substance and drug product. This means that any deviations, mistakes, or problems that are found must be investigated and closed after they have occurred.”
Not only is the process time consuming, but it also means products might be rejected if quality control (QC) problems cannot be resolved. This has the potential to lead to significant revenue loss and product supply constraint.
“The current QC sampling and testing process is cumbersome—it is labor intensive with many potential opportunities for errors in sampling, labeling, transporting, storing and testing.”
The biotechnology plant of the future
Having precise control and a deep understanding of the manufacturing process in real time dramatically reduces the amount of time spent in release testing. ILM/RTRT embeds process analytical technologies directly into the control system, closing the loop on process control by bringing critical process parameters and difficult to measure quality attributes directly into the control system. This inline quality monitoring and control enables teams to perform quality review while treatments are being produced, thereby unlocking RTR capability to prevent potentially millions of dollars of finished product from being held in inventory.
“Biopharmaceutical products are time-consuming and expensive to manufacture, so new methods that are faster and less expensive are actively sought. Implementing ILM/RTR will increase speed and decrease costs and will be a quantum leap in automation of the industry.”
Many biopharmaceutical plants are already actively considering implementation of ILM and RTR, positioning it as a key driver of competitive advantage in the future. According to a survey of members of the BioPhorum Adoption of ILM/RTR Team, nine of eleven organizations already had a roadmap or vision for the adoption of ILM and RTR. While there are still hurdles, such as meeting the required technology, meeting regulatory requirements, and the complexities of developing a business case, they will be overcome.
“These findings show the industry support for this publication and the eventual playbook for adoption of ILM/RTR.”
Getting started
Implementing ILM/RTR will be different for every plant, as it is highly dependent on control architecture and organizational culture. However, while strategies may differ, there is one key constant,
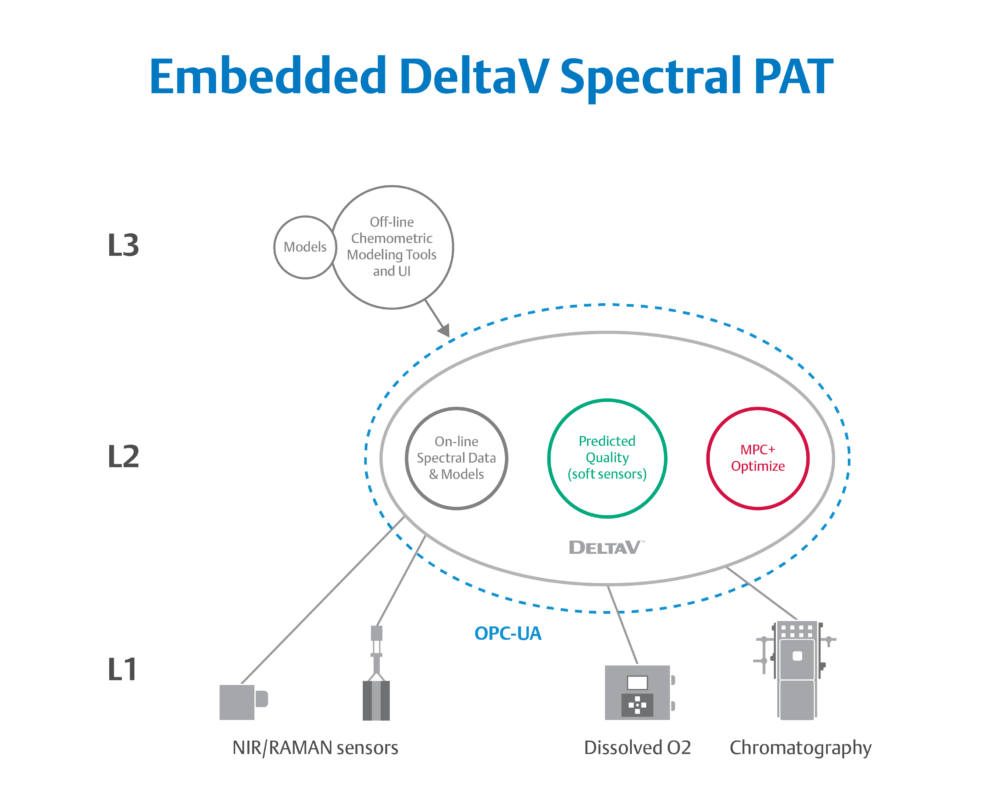
ILM components such as Emerson’s DeltaV Spectral PAT bring real-time quality testing into closed-loop control.
“Regardless of the chosen strategy, the recommended implementation plan is to think big, but start small. Begin by conceptualizing the change, developing a strategic plan, then moving ahead with a pilot, single release or limited rollout. Work with regulatory agencies to move it to completion, then build on the lessons learned from that experience to improve for the next round. Use the lessons learned from the pilot (standardization, reducing complexity, validation, etc.) to move forward to a broader implementation. Along the way, develop stage gates and collect proof of value and return on investment (ROI) data to socialize the concept with stakeholders within the organization. In any strategy, work could be conducted in parallel, keeping the old process while developing the new one alongside it.”
Strategically choosing ILM components such as Emerson’s DeltaV™ Spectral PAT will help life sciences organizations provide a robust architecture that is easy to implement and maintain. Working in a single, integrated environment makes the system more secure and easier to validate and provides ample opportunities to demonstrate ROI.
If you work closely with life sciences manufacturing, be sure to read the paper in its entirety. It is free, and contains much additional content including strategies for implementation, overcoming hurdles and organizational change management, measuring progress, and example case studies.
