Most people have heard the adage that, “COVID changed everything.” However, few people, when presented with that idea, think that any of those changes were positive. But we often forget that in the span of eight short months, the world saw a wide array of innovators...
Speed to Market
Traceability Across the Entire Drug Development Pipeline
Recently, as cell and gene therapies have begun to emerge in the life sciences manufacturing landscape, an important concept has begun to draw attention: traceability. However, it is important to remember that traceability was critical long before today’s most...
Better Treatment—For the Earth, and its People
It can be hard to keep up with the many rapid changes occurring in the life sciences industry these days. In an effort to embrace improved speed to market, sustainability, and quality, life sciences manufacturers are employing a wide range of software and technologies...
Leverage Life Sciences Software to Reduce Time to Market
The days of maintaining paper records across the life sciences development and production pipeline are gone. Today’s regulatory requirements, not to mention the complexity of new treatments, requires organization that can only be provided electronically. However,...
Software Simplifies Technology Transfer
The life sciences marketplace is changing faster than nearly any other industry. Gone are the days when companies focus on a single treatment to drive their entire business. Today’s life sciences manufacturers are challenged to bring new treatments to the marketplace...
BioPhorum’s Roadmap for Speed to Market with In-Line Monitoring and Real-Time Release
Traditional manufacturing practices have served the biopharmaceutical industry well for years, but times are changing and a need for speed to market in life sciences while reducing manufacturing costs is putting a focus on enabling real-time release (RTR)....
Faster Tech Transfer Unlocks Speed to Market
The rapid development of the COVID-19 vaccine has created a dramatic shift in the way the public and life sciences organizations view the manufacturing of pharmaceuticals. In a recent Q&A with Pharma Manufacturing, Nathan Pettus, president of Emerson’s process...
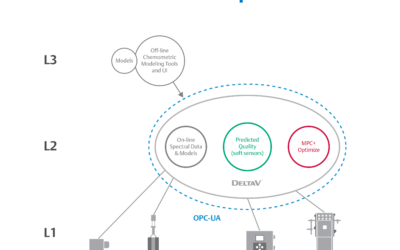
Improve Speed to Market with Closed-Loop Process Control using Spectral PAT
We have witnessed an incredible change the last few years in how life sciences manufacturers deliver treatments. Today’s focus is on fast results, and that means products patients rely on cannot sit on shelves for weeks or months waiting on quality validation. In a...
A Step Closer to Plug-and-Play Engineering with Module Type Package
In the chemical industry, manufacturers often need to change operations or accommodate new products to meet customer demand. However, the extensive integration efforts required to connect manufacturing equipment and automation systems often complicate engineering,...
Modular Plug and Play, MTP Will Enable More Flexible Life Sciences Manufacturing
Modular facilities are a key enabler of speed to market in life sciences. As organizations try to bring new therapies to market faster, those therapies require a wide variety of equipment—many of which are new to the process. These new machines must not only work...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
The opinions expressed here are the personal opinions of the authors. Content published here is not read or approved by Emerson before it is posted and does not necessarily represent the views and opinions of Emerson.