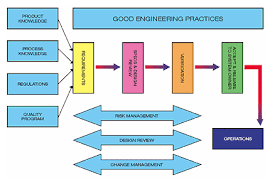
One of the key stages delaying and increasing costs in life sciences projects is equipment validation. In fact, it is one of the elements of project completion that project teams are most eager to compress because the value of every incremental improvement is so...
ASTM E2500
Continued Process Verification in the Process Validation Lifecycle
In 2011, the U.S. Food and Drug Administration (FDA) issued a Guidance for Industry – Process Validation: General Principles and Practices. It highlighted a third validation stage goal of continued process verification (CPV) for: …continual assurance that the process...
ASTM E2500-Early, Targeted Testing Leads to Faster Implementations
Last week, I highlighted Emerson's Heather Schwalje thoughts on Continuous Process Verification per U.S. Food & Drug Administration (FDA) guidelines. In a comment, Pharmaceutical Manufacturing magazine senior editor, Paul Thomas, highlighted a great article, A...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.