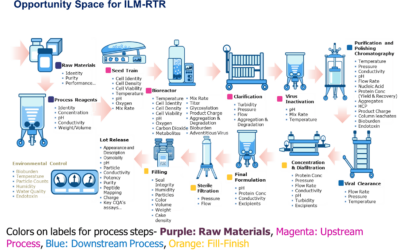
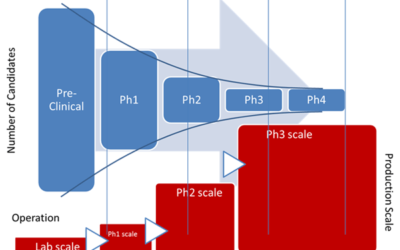
Emerson’s Michalle Adkins will be presenting, Update on BioPhorum’s Roadmap for In-line Monitoring and Real- Time Release at the conference.
biotech manufacturing
Manufacturing Execution Systems in Drug Research and Development
The U.S. Food & Drug Administration (FDA) outlines the drug development process in five steps: Discovery and development Preclinical research Clinical trials FDA review FDA post-market safety monitoring I caught up with Emerson's Zuwei Jin whom you may recall from...
Outlook and Trends for Biotech Industry
What is the 2019 outlook for research & development in the biotech industry? Pharmaceutical Processing magazine explores this question in an article, Outlook 2019: Tech, Data, and Biotech Boom. Emerson's Bob Lenich joined other suppliers to this industry to share...
Best Practice for MES-SAP interface Factory Acceptance Testing
Author: Jonathan Lustri Almost all Life Sciences manufacturing execution system (MES) projects include an interface to SAP, the enterprise resource management system. This interface is strategic in that this is how the business and production are tied together. It...
IIoT in the Life Sciences Industry
When it comes to benefitting from new technologies, pharmaceutical and biotech manufacturers often feel constrained from implementing changes due to possible revalidation regulatory requirements. In this short 1:52 video, Industry 4.0 in Life Sciences, Emerson's John...
Optimizing Media Flow in Biologics Manufacturing
Author: Emily Anderson Flexible facilities are a growing trend in the biopharmaceutical manufacturing industry. Manufacturers increasingly look to produce multiple products and maximize production time. To meet these requirements, an automation strategy must maximize...
Tech Transfer in Drug Development Pipeline and Industry 4.0
Author: Zuwei Jin Among all of the important trends in life sciences industry such as modular plants, integrated modules, out-of-box smart factory, single-use equipment, PAT, and manufacturing execution systems (MES), IIOT has been receiving high interest including...
Technology Trends for Life Sciences Manufacturers
We seem to be fully in the swing of industry conferences. This week is the pharmaceutical and biotech manufacturing industries' INTERPHEX conference in New York City. It: …brings over 11,000 global industry professionals and 650+ leading suppliers together to "Learn...
Data Integrity for Pharmaceutical and Biotech Manufacturers
Update: This post has been updated with a link to the most recent Medicines & Healthcare products Regulatory Agency (MHRA) ‘GXP’ Data Integrity Guidance and Definitions - March 2018 document. For pharmaceutical, biotech and other manufacturers in highly-regulated...
Identify and Resolve Cell Therapy Batch Manufacturing Exceptions
We conclude our series on improving cell therapy manufacturing with a closer look at the review by exception process. In the manufacturing process of cell therapeutics, product cycle times are extremely short and any exceptions occurring during the production process...
Managing the Chain of Identity in Cell Therapeutics Manufacturing
Medicines produced by pharmaceutical and biotech manufacturers are manufactured in many ways. One particular type of treatment, cell therapy, is defined as: …the transplantation of human or animal cells to replace or repair damaged tissue. From a manufacturing...
Successfully Implementing Process Analytical Technology
It's been well more than a decade since the U.S. Food & Drug Administration (FDA) announced a Process Analytical Technology (PAT) approach for pharmaceutical manufacturers in their Guidance for Industry PAT — A Framework for Innovative Pharmaceutical Development,...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
The opinions expressed here are the personal opinions of the authors. Content published here is not read or approved by Emerson before it is posted and does not necessarily represent the views and opinions of Emerson.