In a recently released BioPhorum paper, BATCH DISPOSITION: A shared vision and practical framework for digitized transformation, Emerson’s Michalle Adkins and Debbie Bouwens joined with other experts in the Life Sciences industry to author this work.
pharmaceutical manufacturing
Driving Biotech Manufacturing Performance with Digital Twins
A Genetic Engineering & Biotechnology News article, Beyond Process Development: AI Reshaping Use of Digital Twins, highlights the evolving use of digital twin technology in the Life Sciences industry.
Addressing Pressing Challenges in Life Sciences
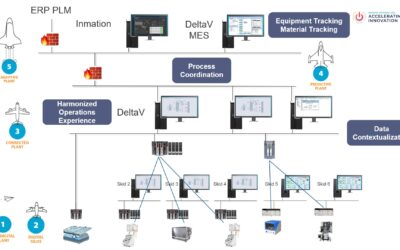
Eliminating silos of data into a comprehensive industrial data fabric enables reshaping the data landscape to provide aggregated, contextualized data for digital transformation initiatives for pharmaceutical & biotech manufacturers.
Award-Winning DeltaV Real-Time Scheduling Software Podcast
David Zhang joins podcast host Jim Cahill to discuss digitalization efforts among pharmaceutical & biotech manufacturers, and the important role that real-time scheduling plays in bringing these therapies to market in a timely manner.
Advancing Life Sciences Automation: Insights from Emerson Exchange 2025
Highlights on a path to more connected sites, systems, and the enterprise. This Emerson Exchange session featured discussions involving foundational connectivity, module enhancements, a scalable data fabric, integrated applications, and an intelligent platform specific to the Life Sciences sector.
Teams of Life Savers Partnering to Accelerate and Deliver Therapies Podcast
Emerson’s Jordan Johnson & Rachel Wright join Jim Cahill in this podcast to discuss challenges in the Life Sciences sector and the role of Emerson Life Savers serve in collaborating closely with them to address and overcome these challenges.
Solutions for Advanced Therapy Medicinal Products
Emerson’s Bruce Greenwald, Christian Berg, and David Gray collaborated to present “Solutions for Advanced Therapeutics” at the Emerson Exchange 2025 Conference.
Unlock the Power of Sustainable & Efficient Measurement Instrumentations in Life Sciences
In today's rapidly evolving world, achieving sustainability has become a pivotal goal, especially in industries like life sciences, where energy efficiency and emission reduction are critical. Emerson’s innovative measurement instrumentations are at the forefront of...
Simplifying Recipe Creation in the Life Sciences Podcast
Melissa Lee & Kebra Tynan join Jim Cahill in this podcast to discuss how Emerson is helping Life Sciences manufacturers address drug development lifecycle challenges with a software-as-a-service (SaaS) based workflow management solution.
Saving the Environment While Saving Lives
The push for more sustainable operations around the globe has touched nearly every industry, and the life sciences are no exception. Manufacturing teams now need to carefully manage waste, energy use, water use, and emissions on top of all their existing...
Enabling Boundless Automation in the Life Sciences Industry
Several of our recent Life Sciences podcasts have featured conversations about the rapid shifts taking place in this dynamic industry. Automation and information management technology are rapidly advancing to help address these challenges. In a recent Emerson...
Life Sciences and Automation Companies Deliver the Future—Together
In the life sciences, bringing treatments to market quicker is no longer a bonus. Today, accelerating the treatment development pipeline is an expectation—arguably a core value—of the world’s most respected pharmaceutical companies. The pipeline for the COVID vaccine...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.