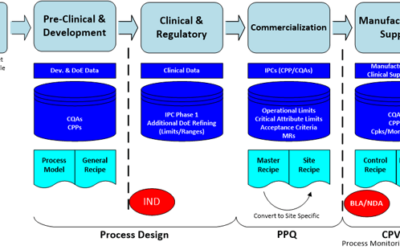
For pharmaceutical and biotech manufacturers, trends in the market place are driving changes in the way they have historically operated their production processes. I caught up with Emerson's Michalle Adkins who shared these trends with me. She identified six major...
Michalle Adkins
FDA Guidance for Data Integrity and Compliance
Author: Michalle Adkins In April 2016, the FDA issued Data Integrity and Compliance With CGMP Guidance for Industry. This is on the heels of a lot of related 483s, warning letters, and more severe actions with respect to this issue. There are some interesting things...
Accurate and Repeatable Measurements in Pharmaceutical and Biotech Manufacturing
A Pharmaceutical Manufacturing magazine study revealed that pharmaceutical and biotech manufacturers were challenged to achieve batch after batch repeatability. I came across a recently published whitepaper, Consistency and Repeatability Through Accurate Measurements....
Life Experiences Shape Consulting Translation Skills
Author: Michalle Adkins Before the holidays, I wrote a blog post, Life Experiences from a Woman in STEM, in the Emerson Exchange 365 Women in Stem group. I talked about growing up on a farm where I gained some early experiences with animals, vitamins, medications,...
Niche-Buster Medicine Information Solutions
Author: Michalle Adkins We are seeing a trend in the life sciences industry moving away from blockbusters to niche busters, population based medicines and even to the extreme of personalized medicines. The Emerson Life Sciences consulting team recently had the...
Improving Data Management, Analytics and Flexibility in Life Sciences
Pharmaceutical and biopharmaceutical manufacturers are challenged on many fronts, such as managing regulatory requirements, operating efficiency, and maintaining the right data required as part of the medicines and products produced. Emerson's Michalle Adkins was...
Optimizing Life Sciences Processes by Improving Data Integration
Pharmaceutical and biotech manufacturers face many economic and technical challenges including reduced prices and diminishing profits, rising research & development costs, increasing regulatory requirements and a greater need for optimized manufacturing...
Data Integrity for Compliance, Audit Readiness and Performance
Pharmaceutical and biotech manufacturers have stringent regulatory compliance requirements. These requirements must balance with the need for improvement and optimization to run safely, efficiently, profitably and in line with the business and quality objectives. In a...
Advancing the Management of Data through Life Sciences Product Lifecycles
From a recent Life Sciences symposium, we looked at issues in the advancement of process intelligence and analytics. Today we'll look at another work session from the symposium, Managing Data Through the Product Life Cycle. The general session was led by Amgen's...
Prototype Manufacturing Operations Management Projects
The ANSI/ISA-95 standard defines models for the information flows between the instrumentation and automation layers up to the enterprise information layers for process manufacturers and producers. Moving up the levels increases the need for people, processes and...
Enabling Release by Exception Manufacturing
The Emerson Exchange conference October 6-10 in Orlando, Florida USA is rapidly approaching. I caught up with Emerson's Michalle Adkins who mentioned that her team of Life Sciences consultants would be highlighting the enablement of release by exception. Although...
What Do You Do for a Living?
Author: Michalle Adkins I often get asked, "So, what do you do for a living?" Boy is that a loaded question. When I say that I manage a group of Life Sciences Industry Consultants for Emerson—that really does not help to answer the question. Hmmm, how do I describe...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
The opinions expressed here are the personal opinions of the authors. Content published here is not read or approved by Emerson before it is posted and does not necessarily represent the views and opinions of Emerson.