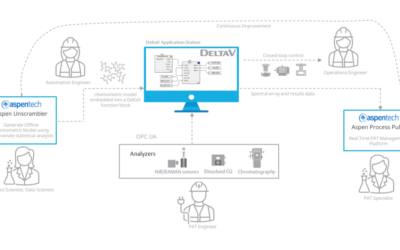
As the landscape of pharmaceutical manufacturing has changed in the last few years, one exciting shift has a distinct possibility of being a game changer in coming decades. Regulatory agencies have increasingly begun to recognize the importance of automation...
pharmaceutical manufacturing
Electronic Batch Reports Support Digital Transformation
Hovione is a contract development and manufacturing company dedicated to helping pharmaceutical customers bring new and off-patent drugs to market. As part of the digital transformation conference stream at Emerson Exchange EMEA 2024, Mariana Brás, senior process...
DeltaV Spectral PAT for Life Sciences Podcast
In this podcast, Emerson’s Jorge Costa and Bruce Greenwald join Jim Cahill to discuss the application of PAT technologies not only within the Life Sciences but across many of the process and hybrid manufacturing industries.
Data Integrity in Life Sciences Podcast
In this Emerson Automation Experts podcast, Michalle Adkins and Hilary Mills-Baker join me to discuss the challenges of achieving the required data integrity and solutions to drive performance improvements.
Achieving Data Integrity, Quality & Compliance in Manufacturing
In a recent webinar, Achieve Data Integrity, Quality & Compliance Across Your Organization, Emerson’s Michalle Adkins and Hilary Mills-Baker share how data integrity means complete, consistent, accurate data throughout its lifecycle.
Driving Operational Improvements with a Rugged PAT Model Infrastructure
At the IFPAC-2023 meeting, Emerson’s Bruce Greenwald and Aspen Technology’s Geir Rune Flåten teamed together to present, Deploying a Rugged PAT Model Infrastructure for Real-Time Monitoring and Control.
Unshackle Innovation with One Click Tech Transfer
Most people have heard the adage that, “COVID changed everything.” However, few people, when presented with that idea, think that any of those changes were positive. But we often forget that in the span of eight short months, the world saw a wide array of innovators...
Life Sciences Digital Maturity – Know Where You Are to Know Where You Can Go
To know where you’re going, you have to know where you are, and operating a life sciences manufacturing facility is no exception. Pharmaceutical processors regularly have to take stock of their technological capacity to keep a finger on the pulse of the four main...
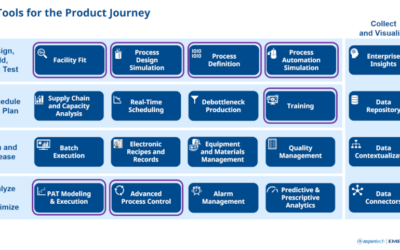
Digital Solutions for Process Development, Tech Transfer and Manufacturing
Emerson’s Michalle Adkins and AspenTech’s Chuck Miller team up to present Robust PAT Solutions to Support Process Development, Tech Transfer and Manufacturing at IFPAC 2023.
Traceability Across the Entire Drug Development Pipeline
Recently, as cell and gene therapies have begun to emerge in the life sciences manufacturing landscape, an important concept has begun to draw attention: traceability. However, it is important to remember that traceability was critical long before today’s most...
Better Treatment—For the Earth, and its People
It can be hard to keep up with the many rapid changes occurring in the life sciences industry these days. In an effort to embrace improved speed to market, sustainability, and quality, life sciences manufacturers are employing a wide range of software and technologies...
Four Pillars of Life Sciences Speed to Market
People are more aware than ever of the many amazing ways new treatments can improve their lives. As a result, life sciences companies are under more pressure than ever to safely deliver new, innovative treatments to market to satisfy customer demand. The key to...
Keep Up to Date With the Latest News and Updates
Follow Us
We invite you to follow us on Facebook, LinkedIn, Twitter and YouTube to stay up to date on the latest news, events and innovations that will help you face and solve your toughest challenges.
Do you want to reuse or translate content?
Just post a link to the entry and send us a quick note so we can share your work. Thank you very much.
Our Global Community
Emerson Exchange 365
This blog features expert perspectives from Emerson's automation professionals on industry trends, technologies, and best practices. The information shared here is intended to inform and educate our global community of users and partners.